In recent reports, it was announced that the National Institute for Health and Care Excellence (NICE) has declined to formalize the acceptance of the use of Vorasidenib on the National Health Service (NHS). Vorasidenib is a targeted therapy that is administered orally for patients with low-grade gliomas. This took place in October 2025 on the grounds of funding, longevity of delaying tumour progression, and its success rates (Brains Trust, 2025; Loney-Evans, 2025).
The initial review by NICE on Vorasidenib took place through its Single Technology Appraisal procedure in November 2024. A subsequent committee occurred in September 2025 to consider evidence from the clinical trial (Loney-Evans, 2025). This led to issuing a draft recommendation in the following month of October 2025, whereby they rejected making the treatment available on the NHS (Loney-Evans, 2025; Brains Trust, 2025).
This has been a massive blow for caregivers, patients, and their families, who had thought it was a promising advance, especially since other options have minimal effects (Loney-Evans, 2025). Some patients like Patient X have described how she was ‘able to do much more’ and her Magnetic Resonance Imaging (MRI) scan presented a smaller decrease in the tumour size which is why the patient wanted to remain on the drug as it allowed the patient to carry the drug as normal rather than endure the harsh effects of chemotherapy and radiotherapy (Hicken, 2025).
MRI is a type of scan used to detect health issues and assist with treatment planning for diseases, and in this case, brain cancer, where the patient is placed in a strong magnetic field and high radiofrequency radiation. Chemotherapy is a treatment that is used to kill cancer cells, whereas radiotherapy kills cancer cells using radiation.
Another patient, Patient Y, reported experiencing fewer seizures and treatment delays (Hicken, 2025). Seizures are abnormal brain electrical activity, causing loss of consciousness, headaches, and loss of movement control.
Stakeholder decisions will conclude on 4th November 2025, followed by the formal NICE committee meeting decision on 20th November 2025 (Brains Trust, 2025). The outcome was initially to be announced at Christmas 2025, and, if approved, was to be implemented within three months of the final approval date (Loney-Evans, 2025). New reports indicated that this has been delayed and rescheduled till after April this year. The decision made by NICE is temporary and not final. It has been estimated that this is the standard procedure performed by NICE, as 84% go on to be approved after further review (Loney-Evans, 2025).
On the contrary, the current decision by NICE does not affect patients who are already on Vorasidenib before the draft guidance was published, nor on a clinical trial patient programme in a pharmaceutical company (Loney-Evans, 2025).
On a global level, many countries have approved the use of Vorasidenib. To date, the United States of America (USA) has granted designation by the Food and Drug Administration (FDA) in March 2023. The FDA is the American Health Authority that prioritised Vorasidenib in February 2025 and later approved it on 6th August 2025 (Loney-Evans, 2025).
In Europe, the European Medicines Agency (EMA) categorised Vorasidenib as a designated orphan medicine in 2023. Orphan medicines are medically used for diagnosis, prevention, and treatment. On the contrary, they have conditional marketing authorisation to ensure medicines are fast-tracked and to meet the unmet medical needs (Loney-Evans, 2025). Moreover, it can be compassionately used for unauthorised medicine outside a clinical study (Loney-Evans, 2025). In February 2024, the EMA rapidly progressed the assessment, which commonly takes 150 days compared to the standard wait of 210 days. The marketing authorisation was approved on 17th September 2025 (Loney-Evans, 2025).
Moreover, in the same month, Vorasidenib was given marketing authorisation in the UK on 16th September 2025 to Les Laboratoires Servier, where it can be used for private healthcare in the UK by the Medicines Health Regulatory Agency (MHRA) (Loney-Evans, 2025; Medicines Healthcare products Regulatory Agency, 2025). On the contrary, not all can afford or sacrifice their means for such treatment.
Ultimately, there is a dire need to ensure equitable access to innovative therapies, allowing real progress. Around 300 people in England are eligible for Vorasidenib (Hicken, 2025). Charities like The Brain Tumour Charity (2025) have committed £60 million since 2015 to driving novel treatment and improving quality of life.
The aim of this article is to present insight into what Vorasidenib is, the type of brain cancers it is prescribed for, and ultimately, the INDIGO clinical trial that observed patients with and without the Vorasidenib arm, and evaluate the reasons why NICE has declined.
What Is Vorasidenib?
Vorasidenib is a drug that is administered orally. It is suitable for patients aged 12 years and above with low-grade gliomas that have mutations of the protein enzyme isocitrate dehydrogenase (IDH) genes (Brains Trust, 2025; Hickin, 2025; Medicines and Healthcare products Regulatory Agency, 2025).
The recommended dose is dependent on body weight. If the patient is less than 40 kg, they should take 20 mg once daily. On the other hand, if the patient’s mass exceeds 40 kg, the ultimate dose is 40 mg daily (Medicines and Healthcare products Regulatory Agency, 2025). It may be offered after surgery, where it is performed on a small scale, a biopsy, or on a larger scale, a sub-total resection or gross total resection (Mehta, 2025).
Low-grade gliomas are commonly characterised by diffuse brain infiltrations. This makes it harder to treat with current treatments of surgery, radiation, and chemotherapy, and can progress to high-grade (Bombino et al. 2024; Mehta, 2025; Cohen, Hokmen, and Colman, 2014). Diffuse Gliomas are categorised by grade: low-grade are grade II, anaplastic are grade III, and the advanced level of glioblastoma is grade IV.
Most low-grade gliomas have a different glial origin of WHO grade 2 or 3; they commonly affect adults under 50 years of age (Brains Trust, 2025). Some gliomas arise in star-shaped cells called astrocytes (70%), whereas others form in oligodendrocytes, another type of glial cell, and form oligodendroglial tumours (9%). Oligodendrogliomas are further classified as classic and mixed with astrocytes (oligoastrocytomas) (Cohen, Hokmen, and Colman, 2014).
The introduction of genetic technologies has enhanced our understanding of identifying genetic mutations in glioma patients. This led to the introduction of IDH and other genes in a high percentage of patients with low-grade glioma. IDH1 mutation and p53 mutation arise in Grade 2 astrocytoma and develop secondary glioblastoma. IDH1 mutation followed by a deletion of the 1p19q gene leads to Grade 2 oligodendroglioma. The deletion of the PTEN gene and amplification of the EGFR gene progress to primary glioblastoma.
There is Baculovirus inhibitory of apoptosis repeat-containing 5 (BIRC), otherwise known as Survivin, which is a gene highly expressed in low-grade glioma patients and considered a biomarker as it inhibits apoptosis (cell death) by preventing enzymes caspase 3 and 7, and helps with cancer cell divisions (Yang et al., 2025).
Normally, IDHs are important enzymes involved in metabolism, particularly the Krebs’ cycle, glutamine, the formation of lipids (lipogenesis), and redox regulation for growth and survival (Han et al., 2020; Loney-Evans, 2025). IDH1 resides in the cytoplasm of cells, where chemical reactions take place, and in peroxisomes. Peroxisomes are small membrane-bound structures found in the cytoplasm and function in lipid metabolism. IDH2 and IDH3 are found in the mitochondria, in particular, the matrix (Han et al., 2020). The mitochondria are where respiration takes place to form energy.
Alpha-ketoglutarate is formed when the chemical isocitrate, which is the target substrate for IDH1 and IDH2, combines with nicotinamide adenine dinucleotide phosphate (NADP+) and a metal ion, magnesium, or manganese, joining together. IDH3 catalyses the formation of alpha-ketoglutarate from isocitrate by utilising nicotinamide adenine dinucleotide (NAD+). NAD+ converts to NADH. The frequently mutated active site residue in IDH found in cancer patients is arginine 132 (R132) (Han et al., 2020).
Under disease states, IDH mutations cause the overproduction of 2-hydroxyglutarate (2-HG) rather than alpha-ketoglutarate (Medicines and Healthcare products Regulatory Agency, 2025). Please see Figure 1. The NADPH converts to NADP+. This promotes tumour growth (Loney-Evans, 2025). Vorasadinib blocks IDH1 and IDH2 by crossing the blood-brain barrier to stop the production of 2HG (Bombino et al., 2024). Lowering the production of 2-HG helps enzymes and DNA methylation. Methylation is the addition of (CH3) groups onto histones, proteins found in the DNA, which help stabilise the DNA structure. This is catalysed by a set of enzymes called histone methyltransferases (HMT). Histone methylation helps alter DNA expression by recruiting regulatory proteins.
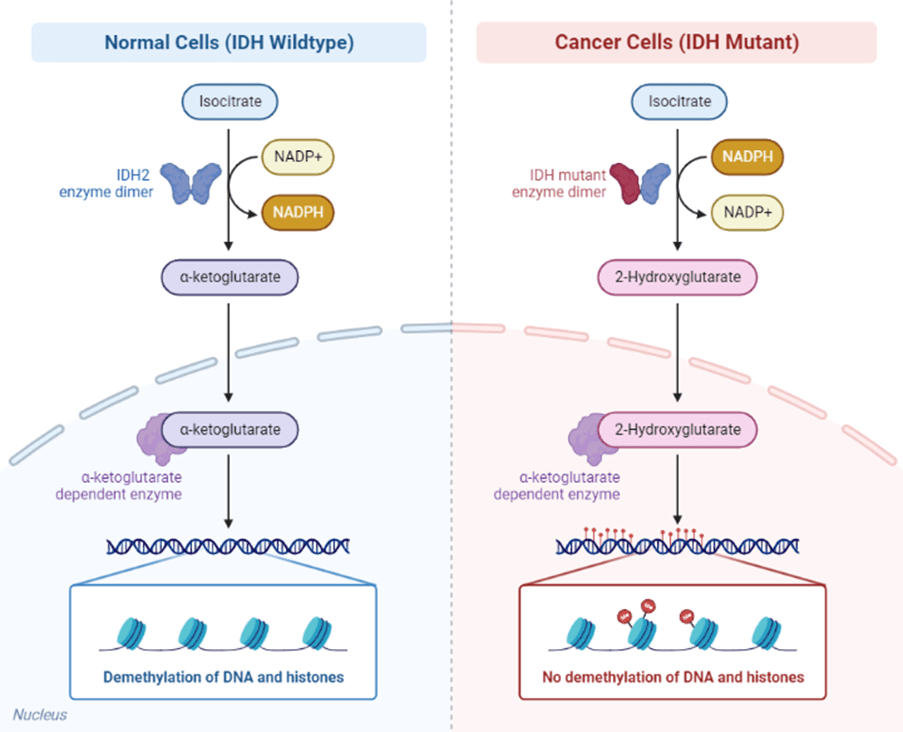
Figure 1: The role of IDH in non-disease and disease states (BioRender, 2025)
About The Clinical Trial
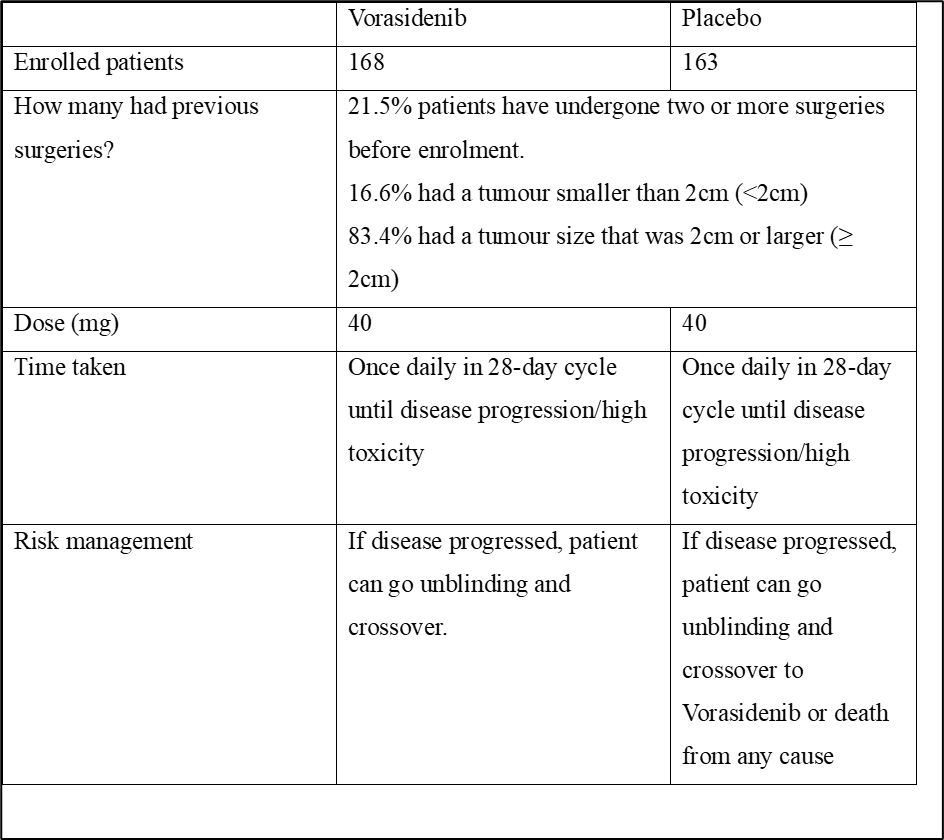
The Phase 3 INDIGO trial is a randomised, multicentred, double-blind, placebo-controlled clinical trial (NCT04164901) (Rosa, 2024). Double-blind refers to neither the patients nor the investigator being aware which one contains the drug and which one does not (placebo). There were 331 patients with IDH1 or IDH2 mutated grade 2 astrocytoma or oligodendroglioma (Medicines Healthcare products Regulatory Agency, 2025; Mehta, 2025). There were 172 patients with oligodendroglioma, and 159 patients had astrocytoma. The characteristics of the patients enrolled are summarised in Figure 2. The median time from the last surgery until randomisation was 2.4 years (Mellinghoff et al., 2023). Patients were from 10 countries worldwide (Brains Trust, 2025). The median age is 40.4 years, with a range of subjects between 16 and 71 years (Mellinghoff et al., 2023). The enrolment began in January 2020 and was completed in February 2022 (Rosa, 2024).
The results suggest Vorasidenib decreased 2-HG by over 90% in resected grade 2 or 3 non-enhancing diffuse glioma (Rosa, 2024). The data were presented at the Society for Neuro-Oncology (SNO) Annual Meeting 2024 (Rosa, 2024). Table 1 and 2 simplifies the outcome of the clinical trial (Rosa, 2024).
Figure 2 The characteristics of patients enrolled onto the study(Rosa, 2024; Mehta, 2025)

Table 1: Overview of the INDIGO trial
(Rosa, 2024; Mehta, 2025; Brains Trust, 2025; Medicines and Healthcare products Regulatory Agency, 2025; Loney-Evans, 2025; Mellinghoff et al., 2023; Food and Drug Administration, 2024)
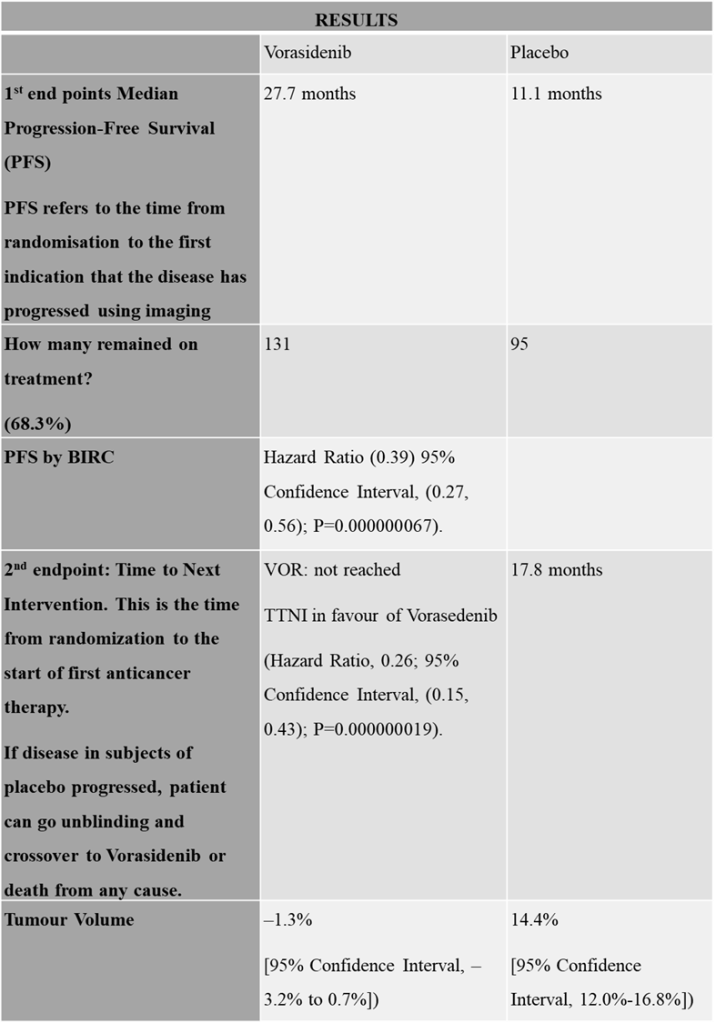
As presented in Table 2, further interventions, chemotherapy, and radiotherapy were delayed, which helped preserve the quality of life (Brains Trust, 2025; Loney-Evans, 2025; Hicken, 2025). Vorasidenib had a longer survival (27.7 months) and longer follow-up time to next intervention (TTNI). It helped decrease the tumour volume significantly more than placebo. Placebo had a Progression Free Survival of 11.1 months and TTNI of 17.8 months.
Table 2: An insight into the results of the INDIGO clinical trial
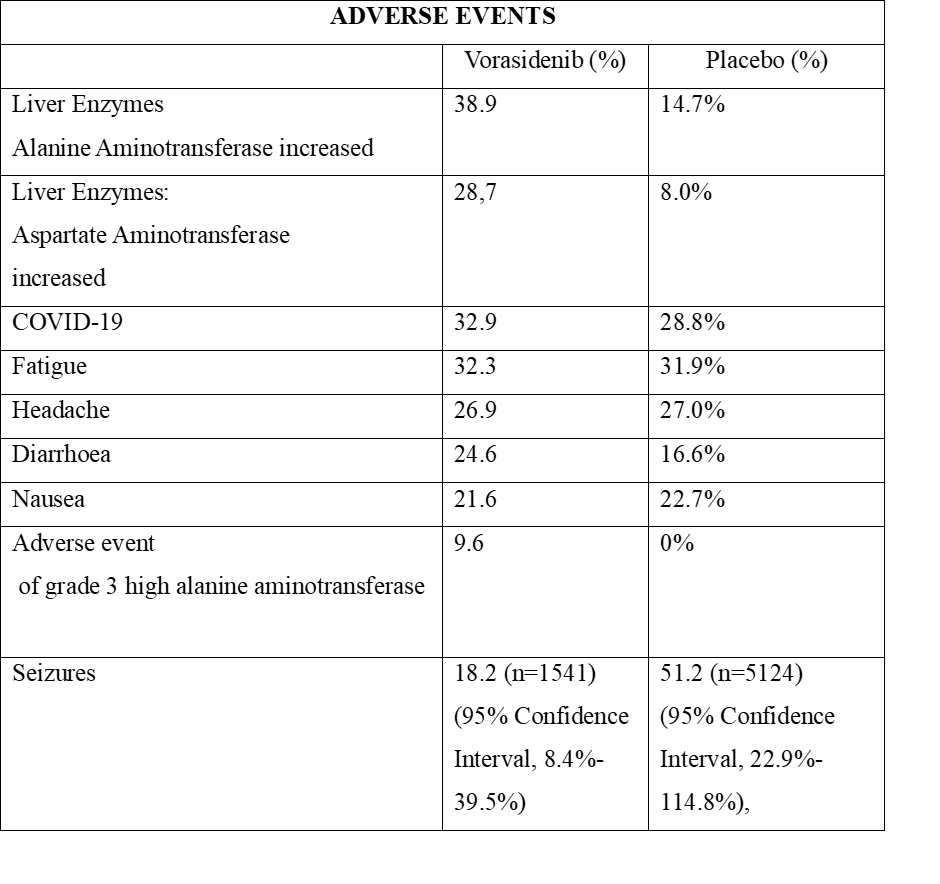
The side effects of the drug Vorasidenib are manageable (Brains Trust, 2025). Key examples are high liver enzymes, fatigue, headache, and nausea as presented in Table 3 (Medicines and Healthcare products Regulatory Agency, 2025). However, other reports suggest that they have long-term adverse effects (Loney-Evans, 2025).
Table 3: An insight into the adverse events of the INDIGO clinical trial
On the other hand. experts in the field of oncology, Mehta (2025), identified several limitations that should not be overlooked. Despite the clinical trial team having enrolled patients who had a previous surgery within the last five years associated with glioma. The number of surgeries conducted has not been indicated, and whether there was a total gross debulking of the tumour, subtotal resection, biopsy, or a combination of all three. This would depend on the size of the tumour and whether there was residual disease (remaining tumour) after surgery, where 16.6% patients initially had tumour <2 cm, and the remaining 83.4% had ≥ 2cm. The criteria for the minimum and maximum tumour size of patients to be enrolled were not indicated. This would be a good comparative measurement in PFS (Mehta, 2025). This may be the reason why NICE has suggested they require further evidence on the long-term outcomes of patients to determine the treatment’s full outcome (Brains Trust, 2025).
Moreover, there was not a wide range of different age groups. There was one patient in the placebo arm who was below 18 years of age and one patient who was older than 65 years. Conversely, there was no one below 18 years of age in the Vorasedenib arm, and two patients were above the age of 65 years. Thus, there was no fine balance between the age groups (Mehta, 2025). Further indicators on patients revealed that 67% had a frontal tumour (Mehta, 2025).
Furthermore, it is important to indicate that Vorasidenib is not a cure. On one hand, it has delayed the progression of tumours but has not stopped the growth, and it is unpredictable when the exact time factor one can subsequently utilise radiotherapy or chemotherapy in patients with low-grade tumours (Brains Trust, 2025). In other words, how long will survival and life be extended?
Overall, the involvement of patients’ perspectives is vital in the decision, as the NHS has been developed for patients, and if the patients see the benefits, then it will be seen as value for money (Brains Trust, 2025). Moreover, every medication has side effects; it is about maintaining the quality of life of cancer patients despite Vorasdenib not being applied routinely (Loney-Evans, 2025).
References
Bombino, A., Magnani, M., and Conti, A. (2024). A Promising Breakthrough: The Potential of VORASIDENIB in the Treatment of Low-grade Glioma. Current Molecular Pharmacology. [online] doi: https://doi.org/10.2174/0118761429290327240222061812.
Brains Trust (2025) NICE’s latest draft guidance on Vorasidenib is not a final decision. Available at: https://brainstrust.org.uk/nices-latest-draft-guidance-on-vorasidenib-is-not-a-final-decision/ (Accessed: 4th November 2025)
Filho, A.M., Znaor, A., Sunguc, C., Zahwe, M., Marcos-Gragera, R., Figueroa, J.D. and Bray, F. (2025). Cancers of the brain and central nervous system: global patterns and trends in incidence. Journal of Neuro-Oncology, 172. doi:https://doi.org/10.1007/s11060-025-04944-y.
Han, S., Liu, Y., Cai, S.J., Qian, M., Ding, J., Larion, M., Gilbert, M.R. and Yang, C. (2020). IDH mutation in glioma: molecular mechanisms and potential therapeutic targets. British Journal of Cancer, [online] 122(11), pp.1580–1589. doi:https://doi.org/10.1038/s41416-020-0814-x.
Hickin, A. (2025). Cambs brain cancer patient disappointed as NHS rejects drug that slows tumour growth. Available at: https://www.cambridge-news.co.uk/news/local-news/cambs-brain-cancer-patient-disappointed-32689619?utm_source=mynewsassistant.com&utm_medium=referral&utm_campaign=embedded_search_item_mobile (Accessed: 4th November 2025).
Loney-Evans, M. (2025). Thanks for helping shape our response to NICE’s draft decision on vorasidenib. Available at: https://www.thebraintumourcharity.org/news/policy-news/help-shape-response-nice-decision-vorasidenib/ (Accessed: 4th November 2025).
Medicines and Healthcare products Regulatory Agency (2025) Vorasidenib approved to treat patients 12 years and older with grade 2 astrocytoma or oligodendroglioma with a susceptible IDH1 or IDH2 mutation. Available at: https://www.gov.uk/government/news/vorasidenib-approved-to-treat-patients-12-years-and-older-with-grade-2-astrocytoma-or-oligodendroglioma-with-a-susceptible-idh1-or-idh2-mutation (Accessed: 4th November 2025)
Mehta, S. (2025) Vorasidenib in Low Grade Glioma. Available at: https://www.bnos.org.uk/wp-content/uploads/2025/06/Vora-in-LGG_FINAL-June-2025.pdf (Accessed: 5th November 2025).
Rosa, K. (2024) Vorasidenib Improves PFS, Shows Tumor Reduction in IDH1/2+ Glioma Available at: https://www.cancernetwork.com/view/vorasidenib-improves-pfs-shows-tumor-reduction-in-idh1-2-glioma (Accessed: 4th November 2025)
Mellinghoff, I.K., Martin, Blumenthal, D.T., Mehdi Touat, Peters, K.B., Clarke, J., Mendez, J.S., Welsh, L., Mason, W.P., Hottinger, A.F., Manuel, J., Wick, W., Riccardo Soffietti, Schoenfeld, S., Zhao, D., Pandya, S.S., Steelman, L., Hassan, I., Wen, P.Y. and Cloughesy, T.F. (2023). INDIGO: A global, randomized, double-blinded, phase 3 study of vorasidenib versus placebo in patients with residual or recurrent grade 2 glioma with an IDH1/2 mutation. Journal of Clinical Oncology, 41(17_suppl), pp.LBA1–LBA1. doi: https://doi.org/10.1200/jco.2023.41.17_suppl.lba1.
Yang, X., Zhang, Y., Duan, F., Li, S. and Wang, G. (2025). BIRC5 Is a Potential Biomarker Associated with Immune System Infiltration in Glioma. Journal of Korean Neurosurgical Society, 68(2), pp.184–201. doi: https://doi.org/10.3340/jkns.2024.0106.
Food and Drug Administration (2025) FDA approves vorasidenib for Grade 2 astrocytoma or oligodendroglioma with a susceptible IDH1 or IDH2 mutation. Available at: https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-vorasidenib-grade-2-astrocytoma-or-oligodendroglioma-susceptible-idh1-or-idh2-mutation (Accessed: 6th November 2025)
Cohen, A.L., Holmen, S.L., and Colman, H. (2013). IDH1 and IDH2 Mutations in Gliomas. Current Neurology and Neuroscience Reports, 13(5). doi: https://doi.org/10.1007/s11910-013-0345-4.
Leave a comment