Cancer Research UK is the UK’s leading cancer charity that holds a prominent stance and multiple pioneering work in developing a better understanding of cancer, diagnosing and treating cancer with the hope of eliminating forms of cancer, preventing people developing it through early detection and awareness of its risk factors and causes and every patient has longer and better quality of life with its world-class technology and global network of clinicians and researchers of all disciplines (Cancer Research UK, 2025a). They incorporate the community through fundraising initiatives to help fund important research projects, making a difference to the lives of patients and their families through effective and targeted treatments. It was established in 2002 but can be traced back to 1902, more than a century ago, when the Imperial Cancer Research Fund was initiated (Cancer Research UK, 2025b). Over the past two decades, cancer survival has increased by two-fold from 1 in 4 people in the 1970s surviving for 10 years or more to 2 in 4 today. The aim of Cancer Research UK is to increase survival rates to 3 in 4 people by 2034.
There are various cancer clinical trials organised by Cancer Research UK for volunteers, whether healthy or cancer patients, to help make a difference and partake in research.
To visit more of Cancer Research UK’s clinical trials
Cancer Research UK (n.d.) Search for clinical trials. Available at: https://find.cancerresearchuk.org/clinical-trials (Accessed: 7th November 2025)
This article discusses a clinical trial in early Phase 1 by the Trial team and Mulholland, P. (2024). The trial runs from December 2023 to December 2026. It examines how patients respond to targeted radionuclide therapy, 123I-ATT001, for brain and spinal tumours that have returned after treatment. Relapsed brain tumours are usually treated with surgery to remove as much of the tumour as possible, followed by chemotherapy. 123I-ATT001 has not yet been approved for medical use by clinical regulators (Trial team and Mulholland, 2024; Cancer Research UK, 2025c).
During surgery, surgeons place a small, soft plastic dome, called an Ommaya reservoir, under the skin of your scalp. Please see Figure 1. After surgery, chemotherapy and other treatments can be given by inserting a small needle through your skin and using a thin tube to deliver the treatment directly to where the tumour is. In this case, the treatment is 123I-ATT001 (Trial team and Mulholland, 2024; Cancer Research UK, 2025c).
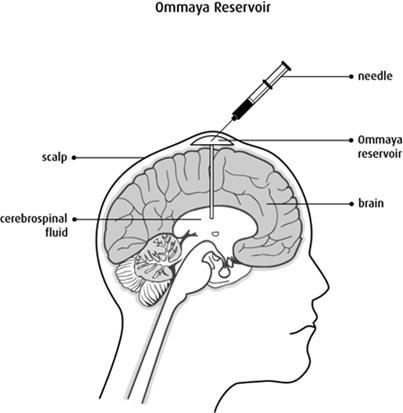
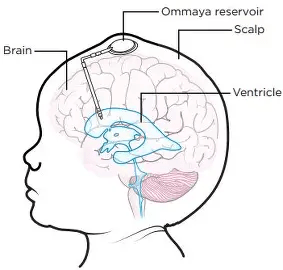
Figure 1: Ommaya reservoir
What Does The Treatment Consist Of?
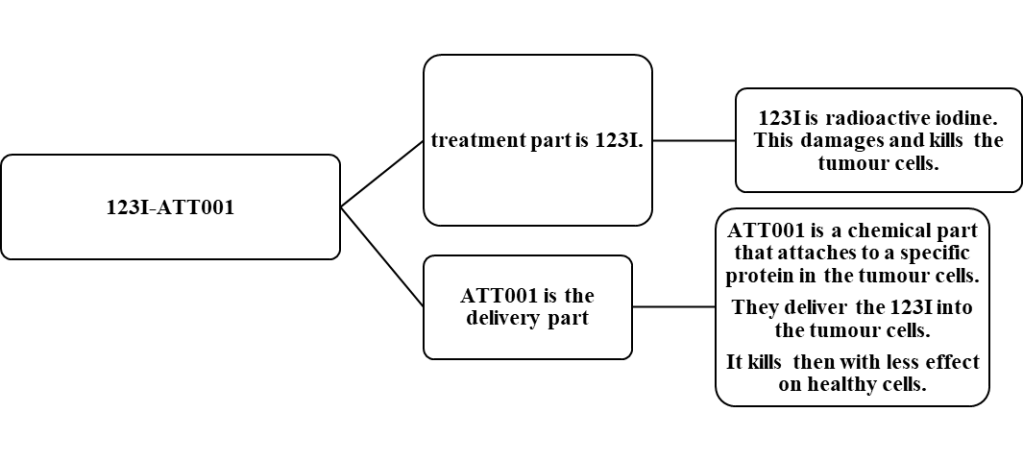
As presented in Figure 2, the origin of 123I-ATT001 consists of radioactive iodine and a chemical that helps with binding to specific proteins in the tumour cells and then undergo apoptosis (Trial team and Mulholland, 2024; Cancer Research UK, 2025c).
The aim is to determine the safety and effectiveness of 123I-ATT001 as illustrated in Figure 3. A series of safety measures was applied. It is vital that trained members of the research and clinical team administer the drug to the patient, take blood, and place the Ommaya reservoir. A radiology specialist reviews the maximum dose and the number of scans acceptable. This helps to lower bruising due to bleeding, infections, and small head bumps, and low radiation exposure (Trial team and Mulholland, 2024; Cancer Research UK, 2025c).

Figure 3: The aim of the clinical study with 123I-ATT001 (Trial team and Mulholland, 2024; Cancer Research UK, 2025c)
How Patients Were Selected For The Study?
Figure 4 illustrates which patients were eligible for the study as a safety net to ethically protect patients from the risk of their health conditions.

Figure 4: A summary of the criteria for including and excluding participants in the 123I-ATT001 study (Trial team and Mulholland, 2024; Cancer Research UK, 2025c)
What Did The Study Involve?
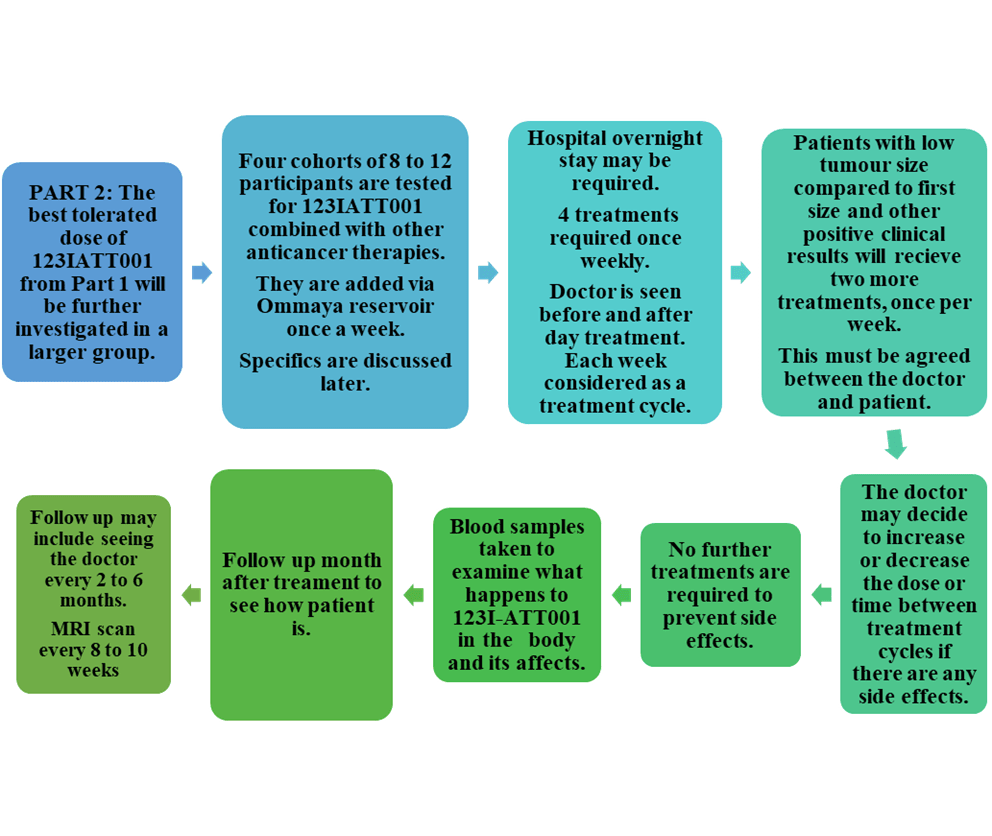
The study was divided into two parts: the first part was to identify the maximal tolerable dose of 123I-ATT001. Please see the summary of the steps involved in Figure 5. Once confirmed, the second part of the study aimed to increase the number of subjects with this dose in combination with other therapies. The effectiveness of 123I-ATT001 was monitored by routine blood tests, cerebrospinal fluid, and measuring the impact on the heart and brain via scanning (Trial team and Mulholland, 2024; Cancer Research UK, 2025c). Please see Figure 6.
Figure 5: Part one of the 123I-ATT001 study (Trial team and Mulholland, 2024; Cancer Research UK, 2025c).
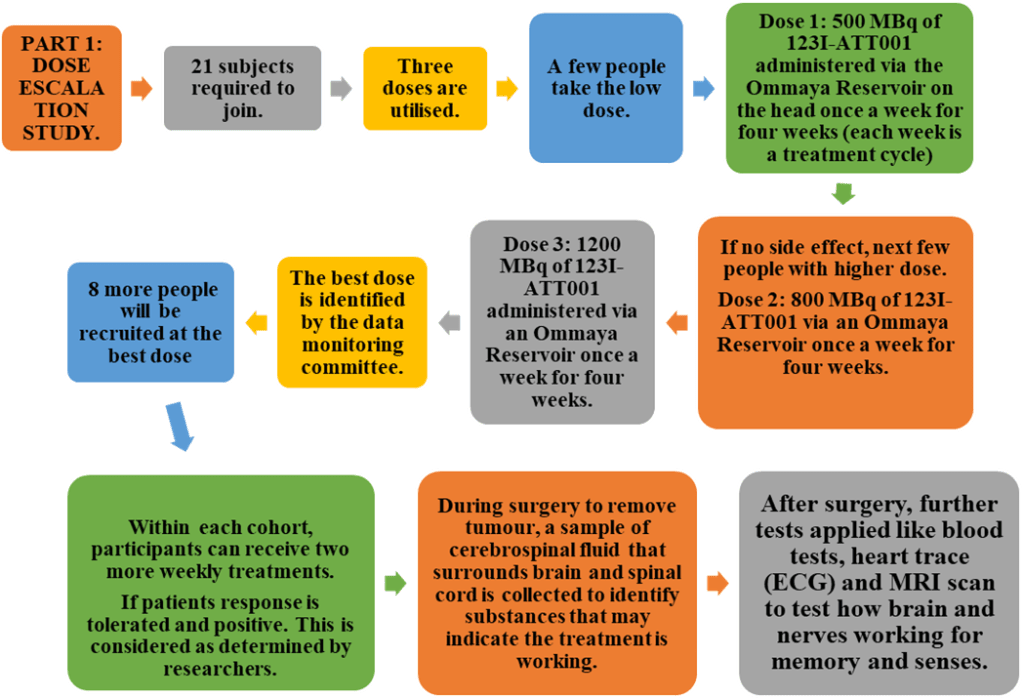
Figure 6: Part two of the 123I-ATT001 study (Trial team and Mulholland, 2024; Cancer Research UK, 2025c).
What Are The Side Effects Of The 123I-ATT001 Study?
With any medication, there are side effects; some are tolerable, but others are moderate or severe and require medical attention (Trial team and Mulholland, 2024; Cancer Research UK, 2025c). The 123I-ATT001 commonly affects the blood, kidneys, liver, and heart, as illustrated in Figure 7. It is important that the doctor is contacted straight away for any apparent signs or symptoms of an allergic reaction. Please see Table 1.
Table 1: A summary of common allergic symptoms post 123I-ATT001
| Signs of minor to moderate allergic reactions | Severe and life-threatening |
| Nose: Sneezing, Itchy, runny, or blocked nose. Eyes: Itchy-red and watery eyes. Lungs: breathless, wheezing, chest tightness, and coughing. Skin: raised, itchy red rash (hives) or dry red cracked skin. Stomach: pain, feeling sick (nausea), being sick, and diarrhoea. | Neurological symptoms: Confusion Lightheadedness Fainting Loss of consciousness. Lungs: difficulty breathing. Blue skin or lips. Swelling of the throat and mouth. |
References
Cancer Research UK (n.d.) Search for clinical trials. Available at: https://find.cancerresearchuk.org/clinical-trials (Accessed: 7th November 2025)
Cancer Research UK (2025a) Our strategy to beat cancer Available at: https://www.cancerresearchuk.org/about-us/our-organisation/our-strategy-to-beat-cancer (Accessed: 15th November 2025)
Cancer Research UK (2025b) Our research Available at: https://www.cancerresearchuk.org/our-research (Accessed: 15th November 2025)
Cancer Research UK (2025c) A study looking at a targeted treatment for glioblastoma (CITADEL-123) Available at: https://www.cancerresearchuk.org/about-cancer/find-a-clinical-trial/a-study-looking-at-a-targeted-treatment-for-glioblastoma-citadel-123 (Accessed: 14th November 2025).
Trial team and Mulholland, P. (2024). Testing 123I-ATT001, a new type of targeted radiotherapy, administered directly to the brain tumour of patients in whom the glioblastoma has returned after previous treatment. http://isrctn.com/. Doi: HTTPs://doi.org/10.1186/isrctn78231121.
Leave a comment