The World Health Organisation has reported that over 70% of cancer deaths globally disproportionately occur in low and middle-income countries (LMIC). The rising global burden is caused by profound inequitable challenges from limited healthcare resources, lack of public awareness of cancers and their prevention, and inadequate training of healthcare staff, which creates a fragile health infrastructure. These limitations can be alleviated by implementing evidence-based strategies, such as prioritizing interventions in cancer services, task-sharing, patient navigation systems, twinning cancer programs, and increasing public awareness through engagement with various communities. All these suggestions from public healthcare researchers will require innovative financing pathways in LMICs to achieve affordability and high-impact technologies.
There are equivocal results on the cancer with the highest incidence and mortality (breast) and the cancer with the least incidence and mortality (thyroid) in 2022, as presented in Figure 1. Most of the top 15 cancers in the global age-standardized rates are gynaecological cancers (cervix uteri, ovary, corpus uteri), genitourinary (prostate, bladder), blood (leukaemia, non-Hodgkin lymphoma), respiratory (lung), breast, gastrointestinal/digestive (colorectum, stomach, oesophagus, oral cavity/mouth, and liver).
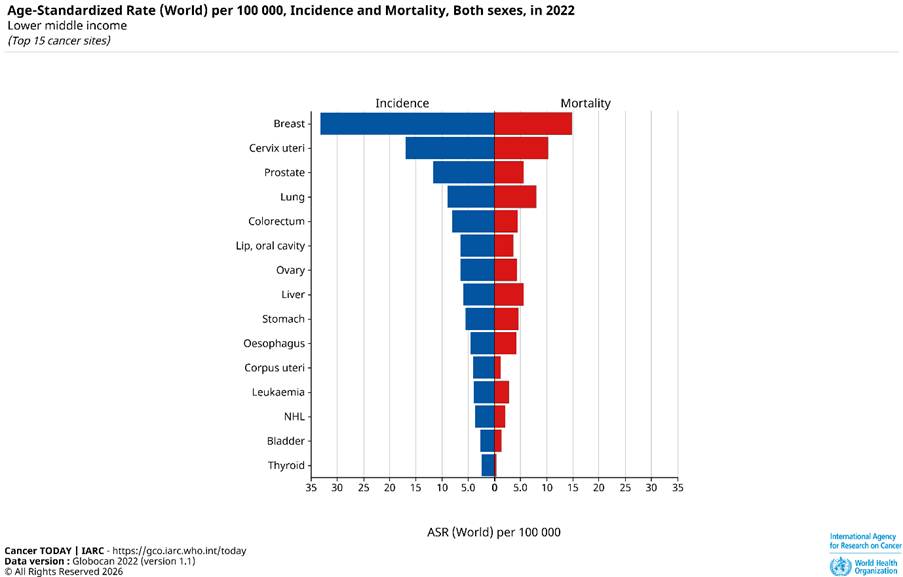
Figure 1: The age-standardized rates for different types of cancers on the incidence and mortality for both sexes in all cancers in 2022.
In Figure 2, the graphical presentation illustrates the age-standardized rate (ASR) per 100,000 of reported incidence and mortality cases among the top 15 LMICs. ASR has been used as a statistical measure to compare health outcomes while accounting for age. Samoa has the highest reported incidence, with approximately ca. 240 ASR per 100,000. Alternatively, Bolivia has the lowest reported incidence, with ca. 140 ASR per 100,000. There is no significant difference reported between Iran, Kenya, Morocco, Vietnam, and Palestine (Gaza and the West Bank), where the reported incidence was ca. 150 ASR per 100, 000.
On the other hand, Mongolia has the highest mortality rate with ca. 180 ASR per 100, 000. In contrast, Morocco and Bolivia had the least mortality rate with a reported ASR of ca. 80 per 100, 000.
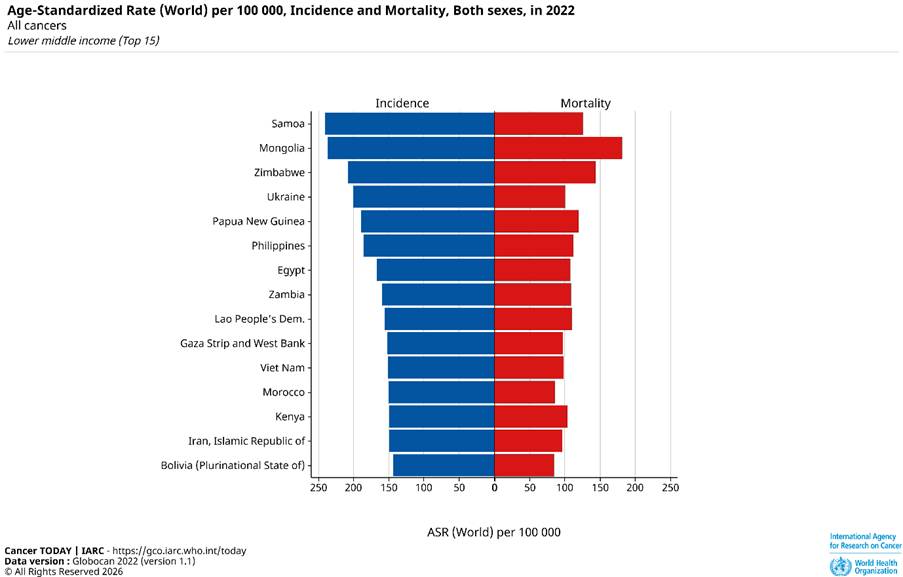
Figure 2: The age-standardized rates for countries with incidence and mortality for both sexes in all cancers in 2022.
Lack of public awareness
Lack of public awareness is the most important factor that contributes to the global burden of cancer cases. This has originated through cultural myths and misconceptions about the causes of cancer that have been speculated from spiritual causes and contagious rather than understanding the connection between modifiable lifestyle behaviours, for instance, smoking, lack of exercise, unhealthy diet, and inadequate measures at factories, mines, industries, and other occupational settings. It has been thought that cancer can be treated through herbal medications alone and faith healing. This has led to a high rise in delays in seeking appropriate medical care, fatalism, and stigma from communities.
This limitation is overcome through community networks, focused interventions, and the application of village health workers to prevent misinformation through culturally appropriate messaging. This has been established through mass media campaigns in print, radio, television, social media, phones, and face-to-face. The development of healthcare packages by the World Health Organisation has drastically reduced the cost of accessing human papillomavirus (HPV) vaccines. More than 85 LMIC countries have been successfully covered. The highest being Rwanda, with more than 90% of adolescent girls covered. Hepatitis B immunisation has also been reported to improve. Oral cancer inspection and tobacco control through increasing tobacco taxes and advertising restrictions. This highlights the successive outcomes through multimedia campaigns and community mobilization to transform the attitudes of cancer awareness and counteract the cancer myths.
Limited Staff and Resources
There is a lack of equipment and treatment regimens in many LMICs due to limited health spending, with total health expenditure remaining <5% of gross domestic product. There are major deficits in access to chemotherapy, surgical tools, radiological equipment, and pathology services across countries.
Integrating radiotherapy is critical for improving local tumour control detection, lowering recurrence risk, and increasing survival. Bamodu and Chung (2024) report that LMICs hold only 5% of global radiotherapy resources. There is a stark contrast in equipment quantity between high-income countries and LMIC. This highlights inequity and geographical disparities. As of 3rd September 2023, the number of radiotherapy machines by income group is high income (11,656), upper middle income (4,693), low middle income (LM), low income (L), and temporarily unclassified (NC) (Bamodu and Chung, 2024).
This has been profoundly discovered in many parts of Africa and Asia where LMIC healthcare systems face daily challenges of radiotherapy shortages and limited staff. Amongst the examples, in 2016, Nigeria had only four radiotherapy centres serving more than 200 million people. Kenya has one public radiotherapy machine and one oncologist per 500,000 patients. Ghana had no radiotherapy machines until 2016, when the first machine was installed, but demand outstrips capacity, resulting in prolonged waiting lists. Other countries reported were South Africa and Taiwan. In addition, countries such as Ethiopia and Malawi still lack radiotherapy in many of their healthcare centres.
This limitation can be addressed through regional cooperation to lower the cost of radiotherapy equipment. Establishing national cancer centres allows countries to share machines. This approach maximises technical expertise, training, and sustainable capacity. The East Africa Radiotherapy Project is a key example, connecting Rwanda, Malawi, and Ethiopia. Another is the Medical Physics Network, which links Nigeria, Tanzania, and Ghana.
Moreover, long turnaround times for pathology services increase the time to report results, assess biopsy samples, and diagnose after symptom onset, and delay the start of treatments. Moreover, LMICs also face shortages of other imaging tools, including magnetic resonance imaging (MRI), ultrasound, computed tomography (CT), endoscopy, Positron emission tomography (PET), and other advanced imaging technologies. This delays detection and provides appropriate staging for cancers that are prevalent, e.g., gynaecological, breast, colorectal, and prostate cancers. This leads to increased scrutiny of advanced disease presentations and poor follow-up.
Furthermore, there is a surprising lack of access to basic pain relief, out-of-stock morphine, and palliative care. Countries like Uganda have 0.5% of the opioid pain medications they require.
Another restraint is the presence of population-based screening programmes. It has been reported that 5% of women in India and sub-Saharan Africa have access to cervical cancer (CaCx) screening programmes. This is conjoined with poor follow-up screening efforts. National mammography screening is available in upper-middle-income countries in South America (Brazil, Mexico), the Far East (Malaysia), and Turkey. On the contrary, other Far-East countries, like China, have an extraordinary level of screening programmes and technology on a broader scale for colorectal, cervical, and breast cancers.
The plague of inadequate resources and equipment can be overcome by innovating financing mechanisms, where the introduction of the Global Task Force on Radiotherapy for Cancer control can help target priority investments, maintain political commitments, and leverage technical expertise. Clinton Health Access has proven to lower medication costs. Ghana’s National Health Insurance Scheme can increase the number of patients receiving free cancer care, particularly among poor and vulnerable groups. Telemedicine can help improve efficiency and appointment access. Collectively, these measures can help prevent cancer-associated expenditures.
Moreover, targeted screening interventions can improve early detection rates, with countries like Nigeria investing in population-based mammography screening in capitals to inform scale-up. More than 500 middle-level healthcare providers in Malawi were trained to produce cervical cancer screening using visual inspection with acetic acid.
Distance travelling
There is a lack of cancer-specialist centres in remote villages and the means to transport patients over long distances. This is compounded by the presence of understaffed and under-resourced facilities in the main cities. Amongst the improvements made in LMICs is that community health workers can travel to rural communities to teach self-examination and provide referrals. This ultimately led to high screening rates in Nigeria.
Inadequate training of healthcare staff and a scarce staff.
Minimal effective techniques and protocols for training oncology specialists are evident in LMIC healthcare systems. There is also a shortage of staff to provide efficient cancer services, including nurses, surgeons, oncologists, radiologists, pathologists, and other healthcare specialists. It has been reported that there are two cancer specialists for 45,000,000 people in Uganda. There are 34 LMIC that have no cancer specialists. This consequently led to an upheaval of many professionals migrating to high-income countries for better resources, training, career growth, and income. This may explain why many healthcare facilities are underresourced and have long waiting times, which consequently lead to poor clinical outcomes.
This factor can be alleviated through partnerships between LMIC and high-income countries to transfer skills and strengthen cancer services. Many are coordinated by the World Health Organisation and university institutions to provide better training opportunities and enhance clinical skills and mentorship.
Another evidence-based strategy is task sharing, in which middle-level practitioners can provide diagnostic testing, screening, treatment, and palliative care to support underserved LMIC communities and address specialist shortages. Key successes included training nurses to perform cervical cancer (CaCx) screening and cryotherapy, which led to an increase from 3% to 15% in screening coverage nationally. Other nurses and clinicians were trained to do biopsies, pain management, and palliative care through structured training, protocols, and support. In Zambia, non-physician clinicians screened and treated CaCx from 4% to >50%.
Overall, numerous global commitments aim to dismantle inequitable barriers as an ethical imperative to improve access to diagnosis, treatment, and prevention, thereby improving clinical outcomes, lowering recurrence rates, improving the efficiency of cancer services, expanding training, and providing opportunities.
References
International Agency for Research and Cancer (2022) Age-Standardized Rate (World) per 100 000, Incidence and Mortality, Both sexes, in 2022. Available at: https://gco.iarc.fr/today/en/dataviz/bars?mode=cancer&types=0_1&sort_by=value1&populations=104_116_12_120_132_144_174_178_204_222_24_262_275_288_332_340_356_360_364_384_404_417_418_426_478_496_50_504_524_548_558_566_586_598_608_626_64_678_68_686_704_716_748_762_788_804_818_834_84_860_882_894_90&group_populations=1 (Accessed: 16th March 2026).
Oluwaseun Adebayo Bamodu and Chung, C.-C. (2024). Cancer Care Disparities: Overcoming Barriers to Cancer Control in Low- and Middle-Income Countries. JCO Global Oncology, 10(10). doi:https://doi.org/10.1200/go.23.00439.
Leave a comment