Ovarian cancer presents the highest rate of morbidity and mortality due to its non-specific symptoms in the early stages and its silent nature. Since the invention of hybridoma technology in 1975 by Köhler and Milstein to generate murine monoclonal antibodies, a mass effort was made to design new engineering strategies to maintain demand, efficacy, and safe use of monoclonal antibodies to diagnose and treat cancers, autoimmune diseases, and infections. In recent years, there has been ongoing development of targeted therapy in the treatment of ovarian cancer.
A brief overview: what are monoclonal antibodies and hybridoma?
Monoclonal antibodies illustrated in Figure 1 aim to target specific antigens found on target cells (microbes or cancer cells). They are Y-shaped glycoproteins that consist of two identical heavy chains and two identical light chains. Each arm contains a variable region that specifically binds to an antigen. The Fc region determines the type or class of antibody and can bind to cellular receptors or complement proteins. As the name suggests, mono (one) and clonal (clone/copies of a single cell), monoclonal antibodies derive from a single clone of a type of white blood cell called B lymphocytes.
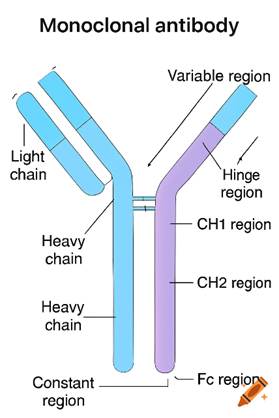
Hybridomas are specialised cells produced in the laboratory by fusing B lymphocytes that generate antibodies from immunized mice (murine) with myeloma cells. Please see Figure 2. Myeloma is a type of cancer cell that has indefinite ability to divide and grow and does not produce antibodies. Alternatively, B lymphocytes have a limited lifespan, where their role is to generate antibodies to recognise and neutralise microbes and cancer cells before they are eradicated by another type of white blood cell called macrophages. Please see Figures 3 and 4.
The fusion between both cells to form a hybridoma enables the dual characteristics of a B cell to produce specific antibodies and the myeloma’s immortal ability to continue growing. The hybridoma product can produce a high volume of a single specific type of antibody. Thus, the specificity and versatility are key attributes in its therapeutic application for many diseases.
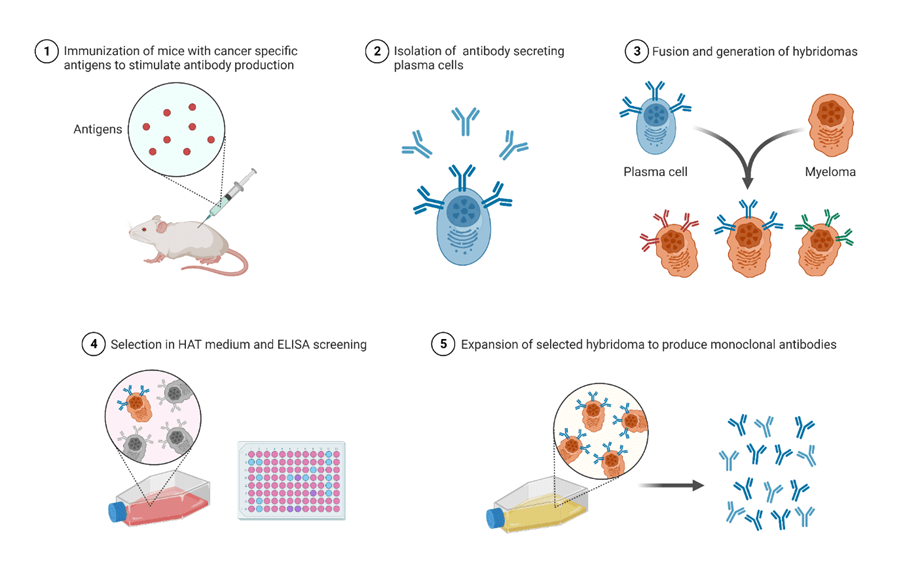
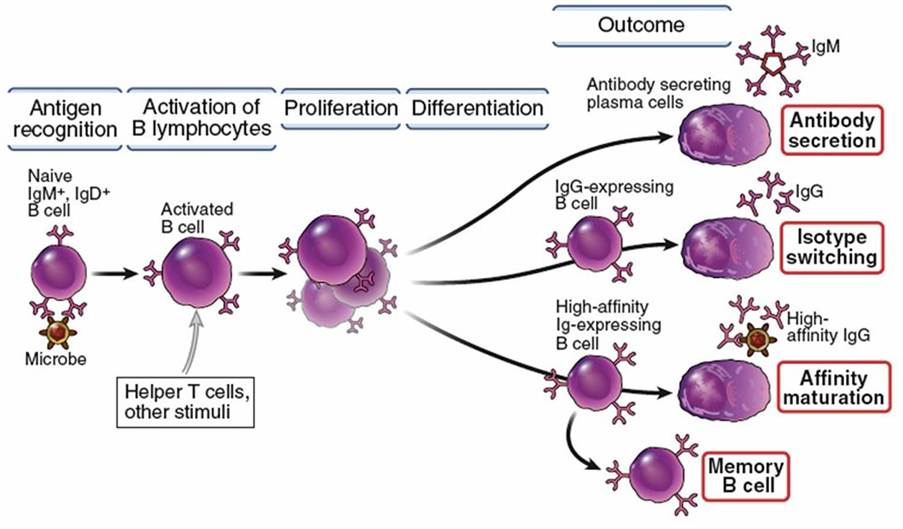
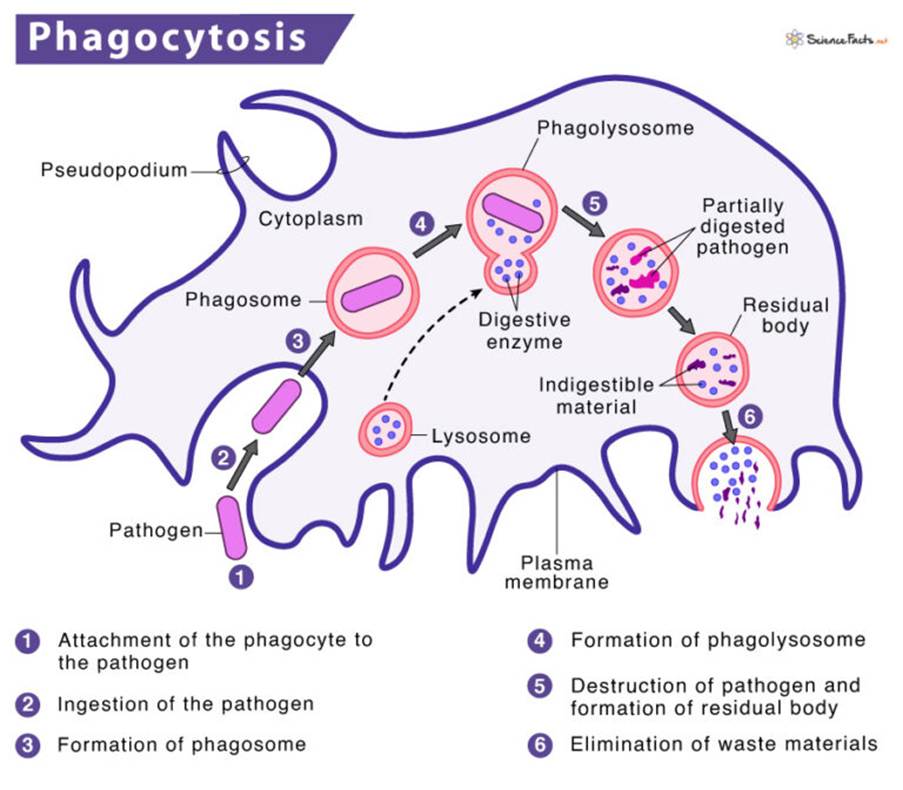
What are new robust methods to produce monoclonal antibodies?
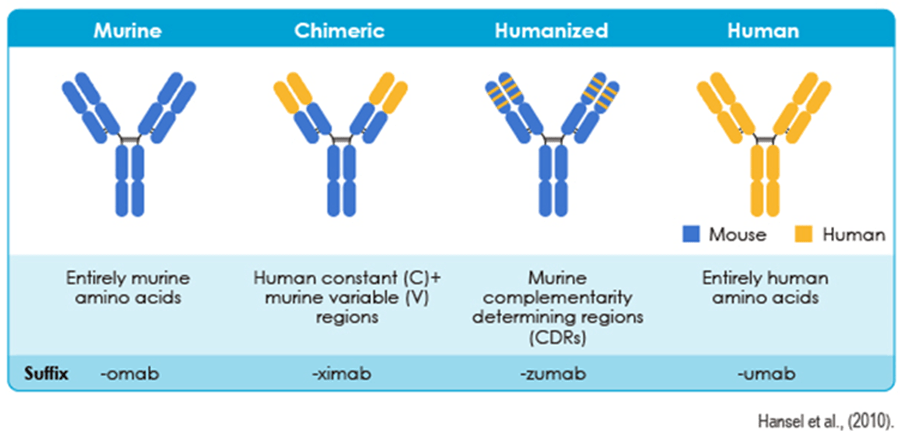
There are several challenges faced when developing hybridomas: cost of production, monoclonal antibodies eliciting immunogenic responses, and personalised medicine designed per patient’s health status. These limitations are overcome by advances in antibody engineering through the following methods: chimerization, humanization, fully human antibodies, and antibody fragments dedicated to improving clinical efficacy, quality of production, lower murine content, and elevated safety (Research Reports, 2025). Please see Figure 5.
Chimeric antibodies have a combination of murine variable regions and human constant regions. Humanized antibodies have lower murine content by replacing it with the complementarity-determining regions (CDR) of a murine antibody on a human antibody. Fully human antibodies are made entirely by humans but can also be made via transgenic mice and phages. Antibody fragments, for instance, Fab, diabodies, and scFv, penetrate tissues and possess lower immunogenicity. They can be engineered to provide specific binding properties and effectors (Research Reports, 2025).
How do monoclonal antibodies work?
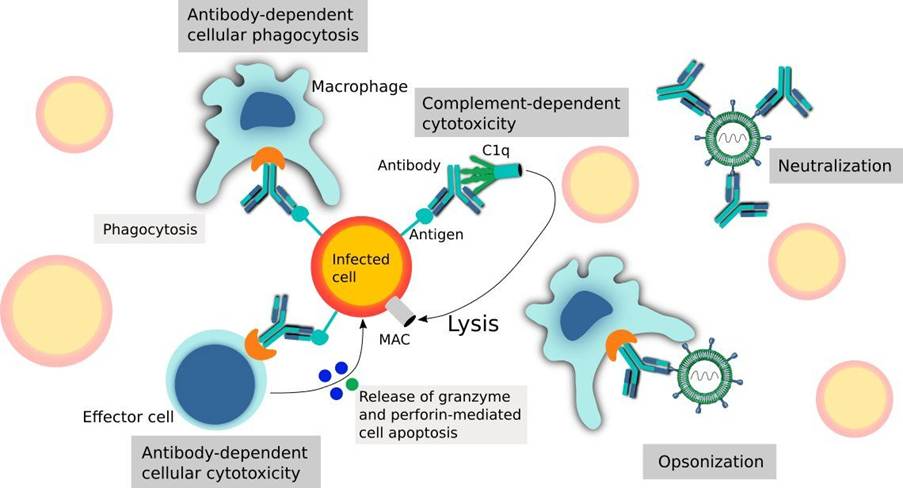
There are various factors that influence the mechanism of action of monoclonal antibodies, and they are dependent on the target antigen, Fc region, and type of disease. They can directly neutralise their target antigen by evading their interaction, recruiting other immune effector cells. Natural Killer cells eradicate target cells to induce antibody-dependent cell-mediated cytotoxicity (ADCC) (Research Reports, 2025). The Fc region of the monoclonal antibodies connects with the Fcγ receptors present on the surface of natural killer cells. This causes the release of cytotoxic granules that kill target cells. Please see Figure 6.
On the contrary, complement-dependent cytotoxicity (CDC) stimulates the complement system, where the Fc region of the monoclonal antibody binds with the complement protein C1q. Please see Figure 6. Complements are functionally linked proteins that aid in the body’s immune defenses when antibodies associate with the antigens. They can break pathogens (disease-causing microbes) or opsonize them. Opsonization is the process by which the opsonin protein attaches to the microbe, rendering it more attractive for macrophages to engulf it. Other functions of the complement system are their involvement in the inflammatory process. Overall, the binding of the Fc to C1q stimulates a complement cascade and the membrane attack complex (MAC), resulting in cell lysis (Research Reports, 2025).
Other modes of action of monoclonal antibodies are their ability to stop immune checkpoints. The role of the immune checkpoints is to regulate the activity of immune cells. An example of how a monoclonal antibody boosts the immune response is to block the inhibitory receptors on T cells more effectively (Research Reports, 2025).
Some monoclonal antibodies mimic the effects of natural ligands, for instance, CD40, to stimulate the B cells to produce antibodies and elicit immune responses. Monoclonal antibodies can also be used as antibody-drug conjugates to deliver cytotoxic drugs to target cells, where the ADC is released, causing cell death (Research Reports, 2025). This illustrates the multiple roles that monoclonal antibodies hold.
The novel development of monoclonal antibodies
Ovarian Cancer Action (2025) reported that the humanized antibody Pembrolizumab has shown effectiveness in patients with recurrent ovarian, fallopian tube, or primary peritoneal cancer who are resistant to platinum-containing drugs. Platinum-resistant ovarian cancer is a profound challenge, where 70% of patients diagnosed with ovarian cancer are at risk of relapse after their first treatment, and platinum drugs have shown no positive effect.
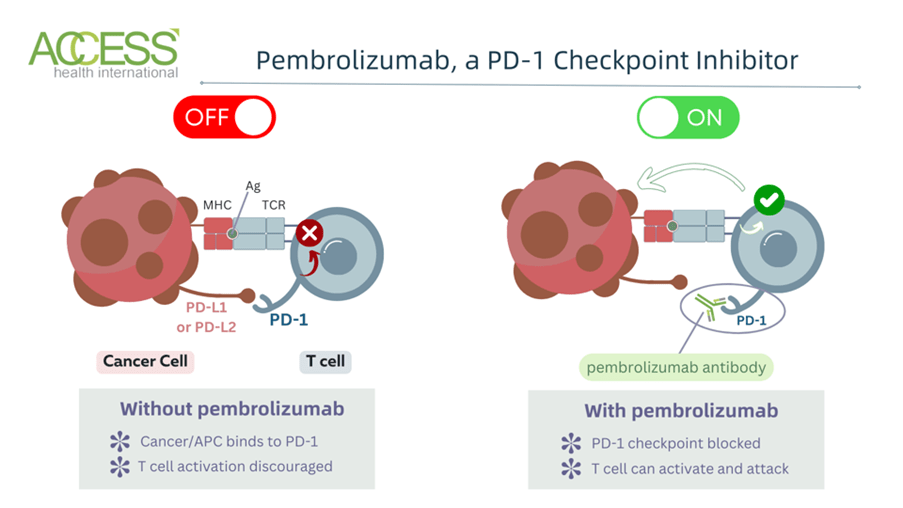
Pembrolizumab is an immune checkpoint inhibitor that targets the programmed cell death protein 1 (PD-1) receptor of T lymphocytes, another type of white blood cell. Please see Figure 7. Actively functioning T lymphocytes in some tumours can eradicate tumour cells, but in other tumours, they have shown no effectiveness because cancer cells can evade the immune system through the PD-1 pathway. The PD-L1 and PD-L2 ligand proteins associate with PD-1 receptors on T cells to inactivate the T cells. Pembrolizumab connects the PD-1 receptor and halts its interaction with PD-L1 and PD-L2. This strengthens and restores the immune response and highlights how PD-1 controls immunity in a cell-intrinsic way. An additional effect illustrates how PD-1 prevents the expression of cytokines. (The University of Edinburgh, 2026). It has previously been shown to be effective in treating lung and skin cancer (Ovarian Cancer Action, 2025).
The large, rigorous phase 3 double-blind and randomised clinical trial, in which neither the doctors nor patients are aware they are receiving the new treatment, reduces the risk of bias and increases the reliability and reproducibility of the results. Every six weeks, Pembrolizumab was administered with weekly chemotherapy, Paclitaxel, with or without Bevacizumab. Bevacizumab is another targeted therapy that blocks the production and growth of blood vessels that provide nutrients for tumour cells to grow and progress. This is achieved by blocking the VEGF protein. The health status of the subjects enrolled in the clinical study has shown improvement and a higher overall survival rate compared to patients who had Paclitaxel alone. More importantly, no side effects were established, which illustrates its safe use (Ovarian Cancer Action, 2025).
In another clinical trial, Pembrolizumab was administered as a monotherapy for patients with renal cell carcinoma (kidney cancer) who are at high risk of recurrence after nephrectomy. Nephrectomy is the surgical procedure that removes the whole kidney or part of the kidney. However, Pembrolizumab illustrated that it lowered the prevalence of recurrence of kidney cancer post-surgery (The University of Edinburgh, 2026).
Newer targeted therapies approved since 2022 are giving people more options, especially when cancer comes back or stops responding to earlier treatments. Biosimilars of the currently approved targeted therapy, Bevacizumab, have been designed to be given as IV infusions and work in the same manner as the original source (Stalvey, 2025).
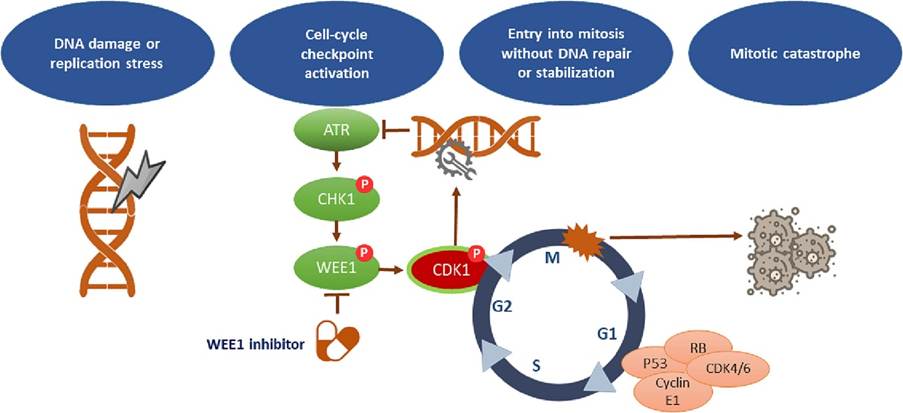
One of the promising targeted therapies is the WEE1 inhibitor. The WEE1 protein is involved in repairing damaged DNA during the cell cycle. Please see Figure 8. Inhibiting the protein can increase the sensitivity of cancer cells to treatment. In Late-phase clinical trials for recurrent ovarian cancer, the WEE1 inhibitor, adavosertib, has shown promising results.
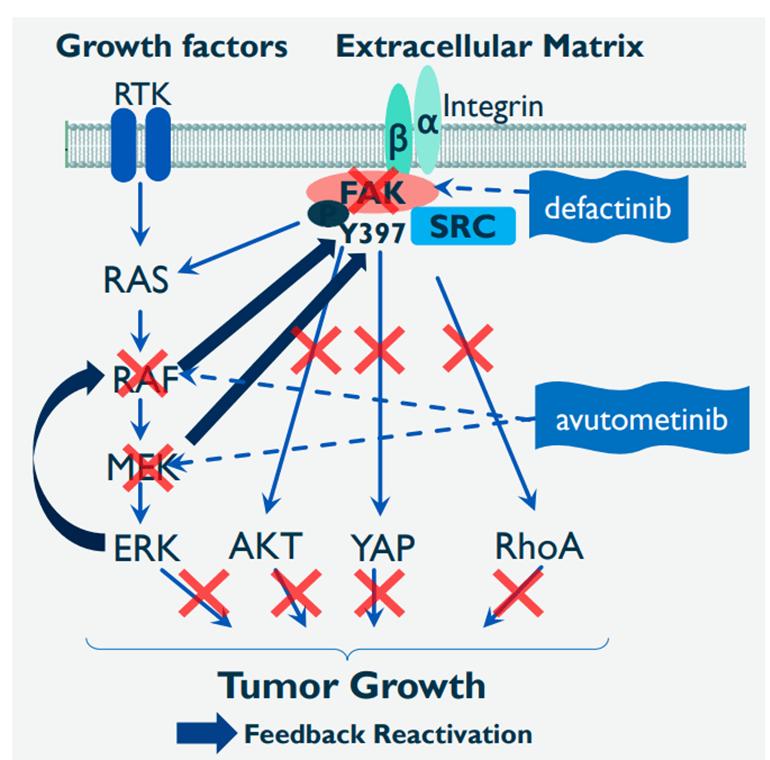
In 2025, the combined targeted therapy, Avutometinib and Defactinib, was approved for the treatment of patients with low-grade serous ovarian cancer with a KRAS mutation who had previous treatment (Stalvey, 2025). There are 15 to 54% of low-grade serous ovarian cancer cases that have KRAS mutations and have clinically presented to increase cancer growth. The combined therapeutic regimens slow the cancer growth by targeting key proteins in the signal transduction pathway (Stalvey, 2025). Please see Figure 9.
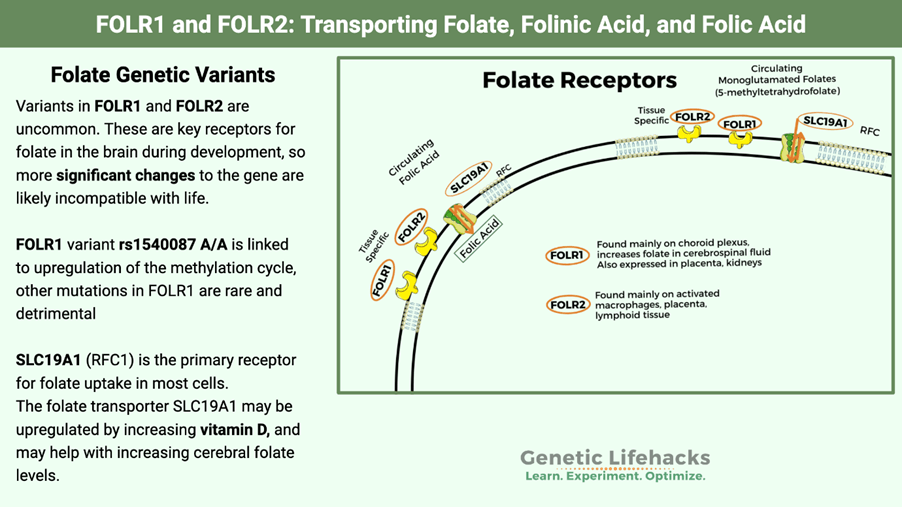
Another targeted therapy, Mirvetuximab soravtansine-gynx, has been recently approved for patients with platinum-resistant ovarian cancers with high levels of folate receptor alpha (FR-alpha) who had one to three treatments previously. The role of folate receptors is to transport folate (folic acid), which has several functions in the brain. During the first three months of pregnancy, it helps prevent neural tube defects like spina bifida. Please see Figure 10. It has been revealed that more than 50% of serous ovarian cancers had high levels of the folate receptor alpha and have shown effectiveness by IV infusion every three weeks.
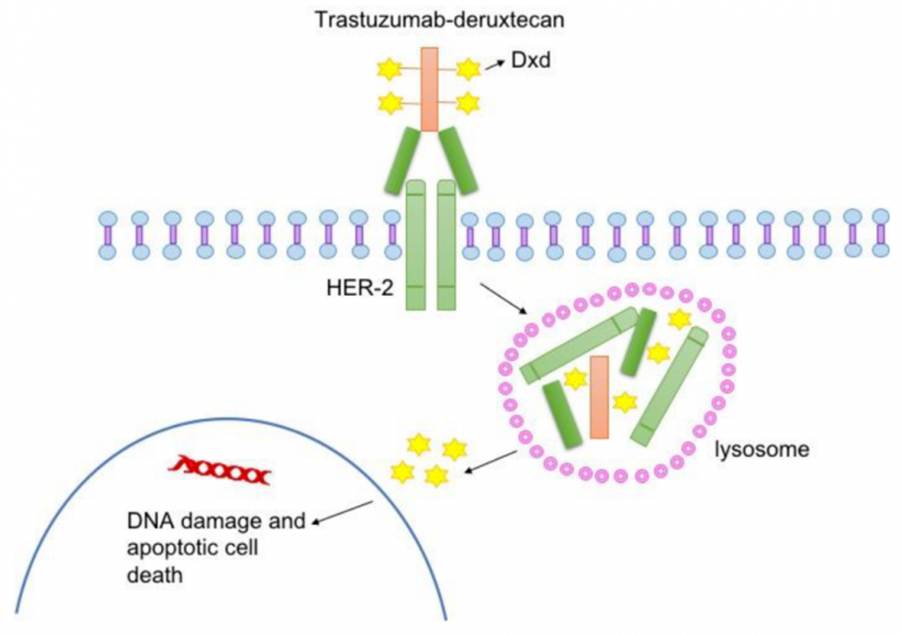
Fam-trastuzumab deruxtecan-nxki is another drug that has been approved to be used in cancer patients who are HER2-positive. HER2 is a protein that facilitates growth, and when mutated, it has been found in aberrant levels in some ovarian tumours. Fam-trastuzumab deruxtecan-nxki attaches to HER2-containing cells and delivers the drug inside the cancer cell, causing cell death. Please see Figure 11. It is administered via IV infusion every three weeks (Stalvey, 2025).
Overall, targeted therapy has promising results for the treatment of patients with ovarian cancer. This humble article highlights some of the key targets established to help increase overall survival rates. Further improvements to overcome challenges from the costs of its manufacturing, potential for immunogenicity, and personalised medicine are in process for a brighter future.
References
Ovarian Cancer Action (2025) New Immunotherapy Combo Offers Hope For Women. Available at: https://ovarian.org.uk/news/new-immunotherapy-combo-offers-hope-for-women/ (Accessed: 17th March 2026).
Research Reports (2025) Monoclonal Antibodies: A Comprehensive Review of Advanced Engineering Strategies, Therapeutic Applications, and Future Directions. Available at: https://medtechnews.uk/research-reports/monoclonal-antibodies-a-comprehensive-review-of-advanced-engineering-strategies-therapeutic-applications-and-future-directions/ (Accessed: 17th March 2026)
Stalvey, K. (2025). Targeted Therapies for Ovarian Cancer: 8 New Treatments and Recent Advances. Available at: https://www.myovariancancerteam.com/resources/targeted-therapies-for-ovarian-cancer-8-new-treatments-and-recent-advances (Accessed: 17th March 2026).
The University of Edinburgh (2026) Kidney cancer drug accepted for use within NHS Scotland and across the UK. Available at: https://institute-genetics-cancer.ed.ac.uk/news-and-events/news-2022/pembrolizumab (Accessed: 17th March 2026)
Leave a comment