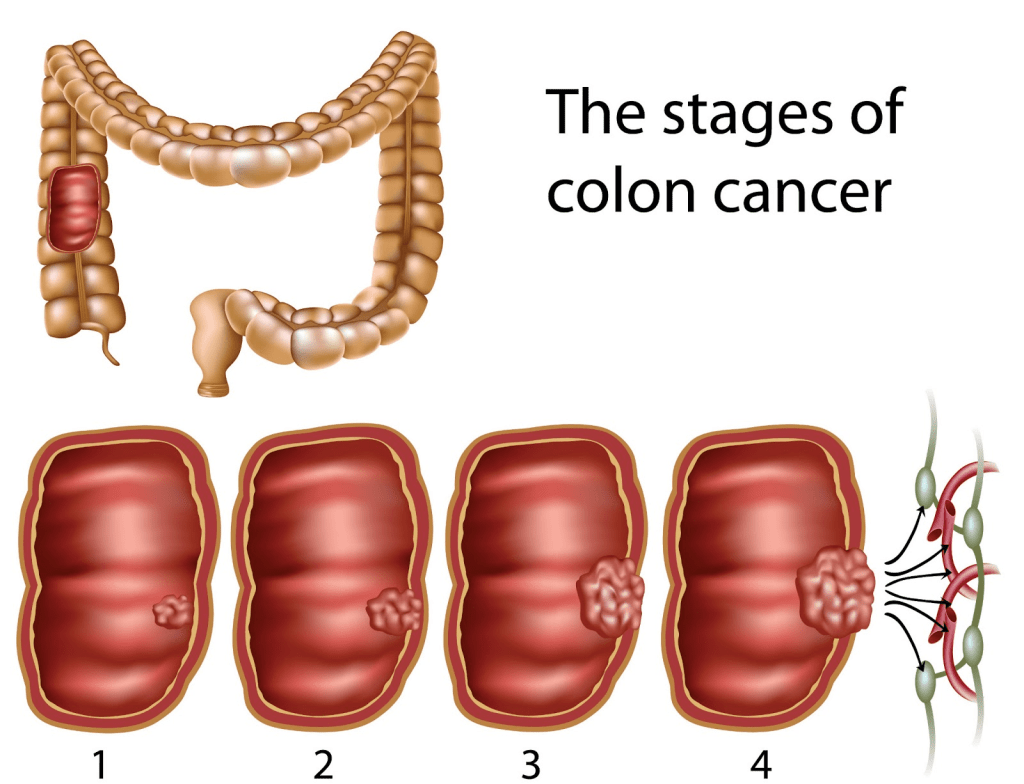
Colorectal cancer is the third most common cancer globally (World Health Organisation, 2026). It is more common in the male population, and the risk increases with age. It is estimated that 35, 000 people are diagnosed with colorectal cancer in England annually (National Institute for Health Care and Excellence, 2025).
The National Health Service bowel screening programme has had a significant impact in detecting patients at risk of bowel cancer, thereby increasing survival rates. Nevertheless, some cases have advanced and are estimated to number 10,000 (National Institute for Health and Care Excellence, 2025). Recently, the National Institute for Health and Care Excellence (2025) revealed that the anti-angiogenic inhibitor Bevacizumab has been approved for the treatment of advanced colorectal cancer in combination with chemotherapy. Now, for the first time, more than 7,000 patients in the UK receive Bevacizumab after the development of biosimilar versions of the same medicine at a lower cost. This presents how Bevacizumab is extensively characterised as targeted therapy, where it is already facilitated in the treatment of gynaecological (ovaries, cervix, fallopian), glioblastoma (brain), renal cell carcinoma (kidney), breast, and non-small-cell lung cancer.
The underlying differences in the genetic, immunological, and environmental origins of colorectal cancer between the proximal (right-sided) and distal (left-sided of the colorectum) essentially require individualised therapeutic approaches. Please see Figure 1. The right-sided colorectal cancer has a lower incidence with a higher prevalence in the female population and exhibits a poor prognosis. Prognosis refers to the patient’s response to treatment over time. Among the molecular characteristics most commonly observed in right-sided colorectal cancer are microsatellite instability (MSI-H), CpG island methylator phenotype (CIMP-H), BRAF, KRAS, and greater mucinous tumours. Several types may arise: CMS1 (immune), CMS3 (metabolic), and the serrated pathway.
On the other hand, left-sided colorectal cancer is more common, especially in males, who are also more likely to have a better prognosis. Its features include CpG island methylator phenotype (CIMP-L) and chromosomal instability. The most common mutations involve TP53, adenomatous polyposis coli (APC), SMAD4, HER2, and epidermal growth factor receptor (EGFR) overexpression. The main types arise from canonical CMS2 or mesenchymal CMS4 subtypes.
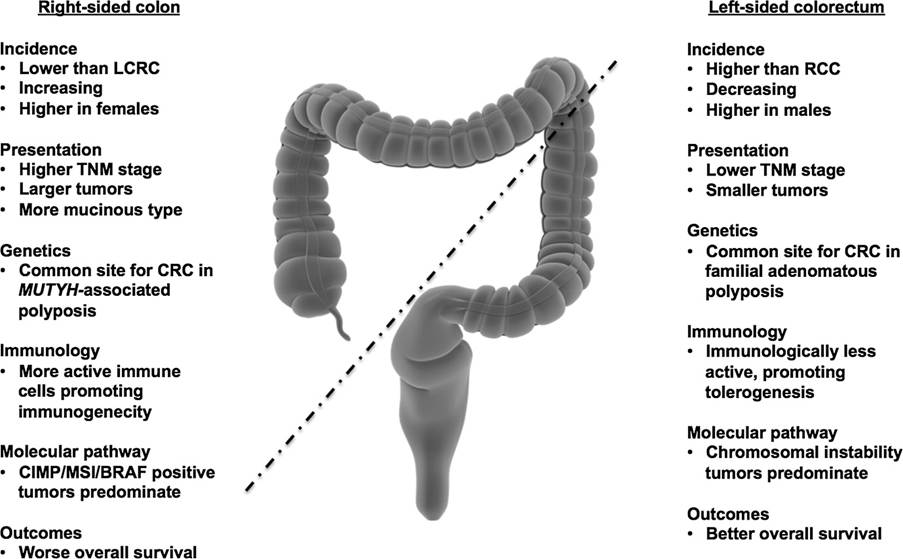
Figure 1: The distinction between left and right colorectal cancer (Lee et al., 2015)
Current Treatment For Advanced Colon Cancer.
Patients with advanced colorectal cancer undergo a systematic approach for treatment whereby cytotoxic chemotherapy is initially used as a first-line chemotherapeutic regimen alone or in combination with other chemotherapies or targeted therapy to increase survival rates for solid tumours. The current NICE guidelines for the management of colorectal cancer are dependent on the pathology status, performance, age, other health conditions, and the patients’ preferences during consultation and reveal the following:
Patients with stage three colon cancer (pT1-4, pN1-2, M0) are treated with combined chemotherapy: capecitabine with oxaliplatin (CAPOX) for three months. If this is not suitable for the patient, oxaliplatin with 5-fluorouracil and folinic acid (FOLFOX) for the duration between three to six months, or alternatively, capecitabine for six months.
Patients with stage 3 (Dukes’ C) colon cancer have a combination of all four chemotherapies: capecitabine, oxaliplatin, 5-fluorouracil, and folinic acid.
Patients with untreated EGFR-positive, RAS wild-type advanced colorectal cancer are treated with the anti-EGFR targeted therapy, Cetuximab, in combination with FOLFOX or alternatively 5-fluorouracil, folinic acid and irinotecan (FOLFIRI) 5-fluorouracil, and irinotecan (FOLFIRI).
Patients with only untreated RAS wild-type metastatic colorectal cancer are treated with Panitumumab targeted therapy in combination with FOLFOX or FOLFIRI.
Patients with untreated metastatic colorectal cancer are treated with Capecitabine monotherapy.
Patients with BRAF V600E mutation-positive metastatic colorectal cancer after previous systemic therapy are treated with Encorafenib with Cetuximab.
Patients with NTRK-fusion-positive tumours are treated with Larotrectinib.
Alternative treatments, for instance, immunotherapy, may benefit patients with microsatellite instability-high (MSI-H) or mismatch repair deficiency (dMMR). The NICE guidelines recommended that Nivolumab with ipilimumab can be used to treat untreated non-resectable advanced cancers with this type of mutation. Pembrolizumab is recommended for advanced colorectal cancers, but is paused if the disease progresses within two years or before.
Relieving symptoms can be established through radiofrequency ablation, cryoablation, radiotherapy, or surgery. Laparoscopy is recommended over open surgery to treat colon cancer when both options are available for patients with incurable advanced cancers who are receiving chemotherapy, and also for patients who do not have any symptoms (asymptomatic).
However, conventional treatment may lead to drug resistance and the development of incurable recurring colorectal cancers. This illustrates the dire need for novel approaches in the management of colorectal cancer.
About Bevacizumab and how it works
Initially, the NICE guidelines did not recommend the targeted therapy Bevacizumab for patients with untreated advanced colorectal cancer, whether in combination with 5-fluorouracil and folinic acid or with oxaliplatin and either 5-fluorouracil, folinic acid, or capecitabine. The main reason revealed is the value for money. However, the development of biosimilars of Bevacizumab has lowered manufacturing costs and expanded patient access to proven therapies.
Moreover, a recent clinical trial revealed that Bevacizumab can be combined with chemotherapy intravenously infused once every two or three weeks, as it can slow down cancer progression and improve overall survival in comparison to chemotherapy alone. The results have indicated that patients with Bevacizumab and chemotherapy had slowed their cancer progression for 9.4 months on average in comparison to 8 months with solely chemotherapy. The overall survival was 21.3 months, compared with 19.9 months with chemotherapy alone. This illustrates the significant impact Bevacimab with chemotherapy had on the prognosis of cancer patients. Hence, NICE has elevated its position to serve as a first- and second-line option when immunotherapy is deemed unsuitable, thereby enabling clinical decision-making (National Institute for Health Care and Excellence, 2025).
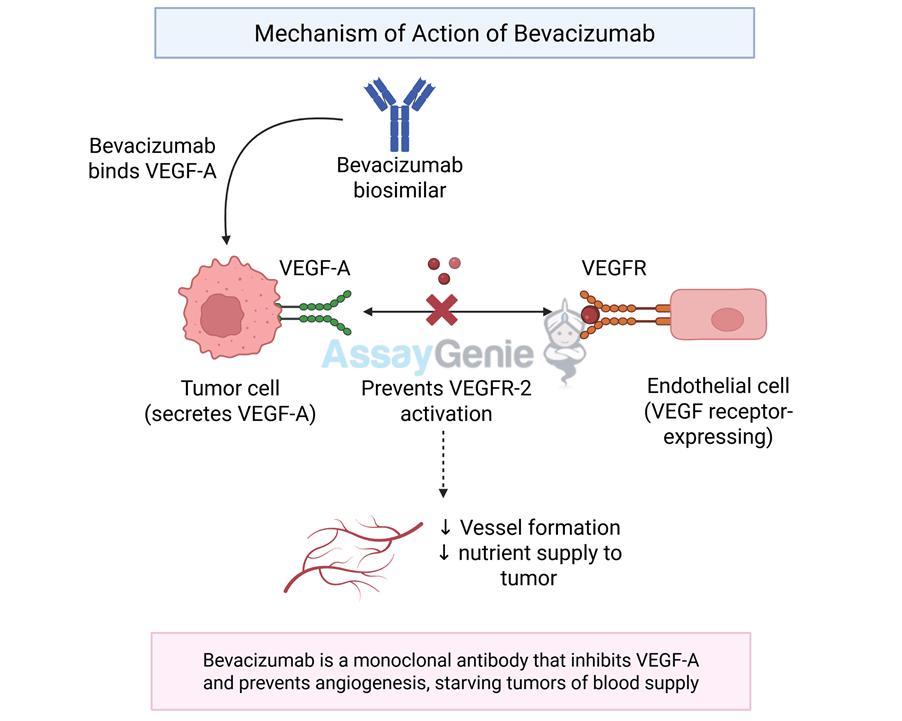
Bevacizumab is an antiangiogenic agent that targets VEGF. VEGF is part of a family of six structurally-related proteins (VEGFA, VEGFB, VEGFC, VEGFD, VEGFe, and placental growth factor) whose role is in the production, growth, and differentiation of the blood vessels (angiogenesis) and lymph vessels (lymphoangigenesis). The predominant angiogenic effects are elucidated primarily by VEGF, one of its receptors, VEGFR-2. As Figure 2 shows, the Bevacizumab biosimilar binds to VEGFA, which is secreted by the tumour cell. This prevents the binding of VEGF-A to VEGFR2 on the endothelial cell lining. This also blocks the formation and decreases the nutrient supply to the tumour.
Overall, the screening and treatment of colorectal cancer are evolving in response to tissue heterogeneity and are influenced by the understanding of the risks and causes of the disease, the type of colorectal cancer (right or left), pathological status, age, other health conditions, lifestyle, and other contributing factors. Colorectal cancer in the early or advanced stages needs to be equitably treated to ensure each patient has the best quality of life outcome, decreasing the symptoms experienced, and resectable or alternative therapeutic modalities.
References
Lee, G.H., Malietzis, G., Askari, A., Bernardo, D., Al-Hassi, H.O. and Clark, S.K. (2015). Is right-sided colon cancer different to left-sided colorectal cancer? – A systematic review. European Journal of Surgical Oncology (EJSO), 41(3), pp.300–308. doi:https://doi.org/10.1016/j.ejso.2014.11.001.
National Institute for Healthcare and Excellence (2021) Colorectal cancer. Available at: https://www.nice.org.uk/guidance/ng151/chapter/Recommendations#management-of-local-disease (Accessed: 19th March 2026).
National Institute for Healthcare and Excellence (2025) Thousands more people with advanced bowel cancer to benefit as NICE approves bevacizumab biosimilars. Available at: https://www.nice.org.uk/news/articles/thousandsmorepeople-with-advanced-bowel-cancer-to-benefit-after-nice-gives-green-light-tobevacizumabafteravailability-ofbiosimilars (Accessed: 19th March 2026)
World Health Organisation (2026) Colorectal cancer. Available at: https://www.who.int/news-room/fact-sheets/detail/colorectal-cancer (Accessed: 19th March 2026)
Leave a comment