Ovarian cancer is one of the most critical health concerns in the female population worldwide. It is the most common gynaecological malignancy, often detected late due to non-specific symptoms at early stages. It has been reported that two of the main bioactive compounds that originate from the Cannabis sativa plant, cannabidiol (CBD) and delta-9-tetrahydrocannabinol (THC), have proven effective against ovarian cancer. Cannabis is a renowned bushy plant that was utilised as a medical treatment for many cancers, namely, glioblastoma, lung, skin, prostate, and breast cancers, and other illnesses, for instance, anxiety disorders and epilepsy. It contains more than 70 different phytocannabinoids, creating a good pharmacological profile (Boggs et al., 2016).
CBD and THC are classified as endocannabinoids; however, CBD is a non-psychoactive compound, whereas THC is. Both are classified as endocannabinoids. Please see Figure 1. There are two primary receptors in the brain: cannabinoid 1 receptors (CB1Rs) and cannabinoid 2 receptors (CB2Rs). CB1Rs are predominantly found in the neurons/nerve cells of the nervous system. The nervous system is subdivided into two divisions: the central nervous system (CNS), which contains the brain and spinal cord, and the peripheral nervous system (PNS), which consists of the peripheral nerves around the bound.
The CB2Rs reside on glial cells, the immune system, and the enteric nervous system (abdomen). The glial cells are non-neural cells of the CNS whose role is in the structural framework, supporting, and insulating the nerve cells. There are some receptors, GPR55 and peroxisome proliferator–activated receptor-alpha [PPAR-alpha], that are stimulated by endocannabinoids but are not formally considered as part of the endocannabinoid system. This illustrates how each type of receptor is specific to elucidate psychotomimetic, immunological, and anti-inflammatory effects. However, CBD binds to both the CB1R and CB2R but acts as an antagonist at these receptors (Boggs et al., 2016).
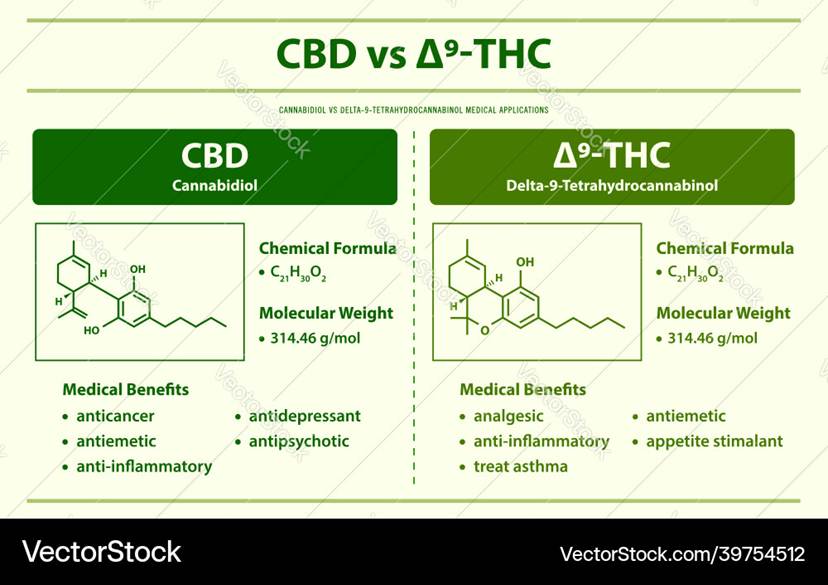
The link between ovarian cancer and cannabis
Recent cellular studies discovered that CBD exhibits many anti-cancer properties in a dose-dependent and independent manner: preventing cell proliferation, inducing reactive oxygen species, evading angiogenesis, and other hallmarks of cancer. However, there were minimal effects on non-cancerous cells, illustrating that CBD and THC are less toxic and tolerable.
The experimental study examined two different ovarian cancer cell lines and one ovarian non-cancer cell line (IOSE80) as a control in vitro. One of the cancer cell lines was platinum-sensitive (A2780), whereas the other was platinum-resistant (SKOV3). The researchers aim to explore their treatment responses towards CBD, THC, and a combination of both on different cell lines that mimic different types of ovarian cancers (Boggs et al., 2016).
The initial experiment involved treating the three cell lines (SKOV3, A2780, IOSE80) with different concentrations of CBD and THC at three time intervals (24, 48, and 72 hours) in a dose-dependent manner. Inhibitory effects were presented in the cancer cell lines (SKOV3 and A2780) at all three time points, with the strongest cytotoxic level at 48 hours. This was determined by measuring the half-maximal inhibitory concentration (IC50). The IC50 is the concentration required to inhibit cells by 50% as a measure of potency. The IC50 concentrations for the cell lines were 4.33 ± 0.11 µM (SKOV3), 5.07 ± 0.37 µM (A2780), and 21.65 ± 1.49 µM (IOSE) when treated with CBD. This illustrates that the SKOV3 cancer cell line was more sensitive to CBD treatment than A2780, though there is a 0.74 minimal difference. In contrast, there is a five-fold difference between the marked potency exhibited by ovarian cancer cell lines compared to the IOSE cell line (Boggs et al., 2016).
Analogous results were established when all three cell lines were treated with THC. The notable toxic level was at 48 h. The IC50 values were 5.75 ± 0.21 µM, 5.92 ± 0.12 µM, and 24.42 ± 2.19 µM for SKOV3, A2780, and IOSE cells, respectively. This indicates both CBD and THC have the ability to decrease cellular proliferation in tumourigenic cell lines more than in non-tumourigenic cell lines. This finding also illustrates how CBD and THC can lower the rate of metastasis of different ovarian cancers, as both were similarly affected.
Moreover, the Chou-Talalay method was performed on all three cell lines to assess their impact on cell death (Fa). Results signified increased cell death synergistically in a dose-dependent manner at equimolar low concentrations for the A2780 cell lines. There were higher fractions of cell death exhibited in A2780 cells than in SKOV3 cells. The SKOV3 cell line required higher Fa values to present the 1:1 ratio of CBD: THC. This highlights some complexity regarding the precise concentration and ratio dependency required to induce synergetic interactions (Boggs et al., 2016).
Furthermore, cellular studies revealed that the cytotoxic effects of CBD and THC greatly reduced the number of cancer cells compared to the control, highlighting their individual impact on colony formation. Alternatively, their effects were amplified when combined in a 1:1 ratio (2.5:2.5 µM), inhibiting clonogenic potential. The most significant difference was with the A2780 cell line ( p<0.0001) (Boggs et al., 2016).
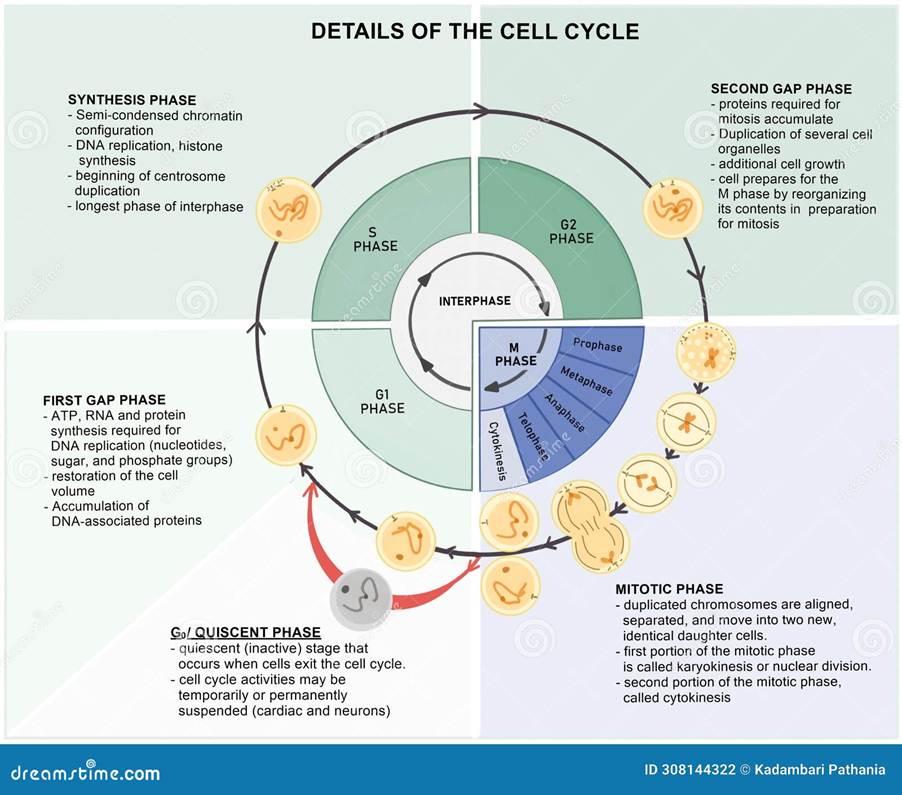
The role of CBD and THC in cancer also affected the cell cycle. The cell cycle consists of three phases. G1 (growth phase), S phase (DNA replication), and G2 (additional growth phase). The G0 phase is the quiescent phase where no action takes place. Please see Figure 2. Boggs et al. (2016) revealed that at 5 μM concentration, CBD or THC solely decreased the number of cells progressing through the cell cycle and placed them in the G0/G1 phase. The number of cells increased in the G0/G1 phase in the combined treatment. In contrast, the control cell line had more cells progressing through the cell cycle, 40% were in the G1/G0 phase, whereas 60% was in the S and G2/M phase. This illustrates how CBD, THC, or their dual effect selectively target the cancer cell cycle to induce arrest rather than the non-cancer cell line (Boggs et al., 2016).
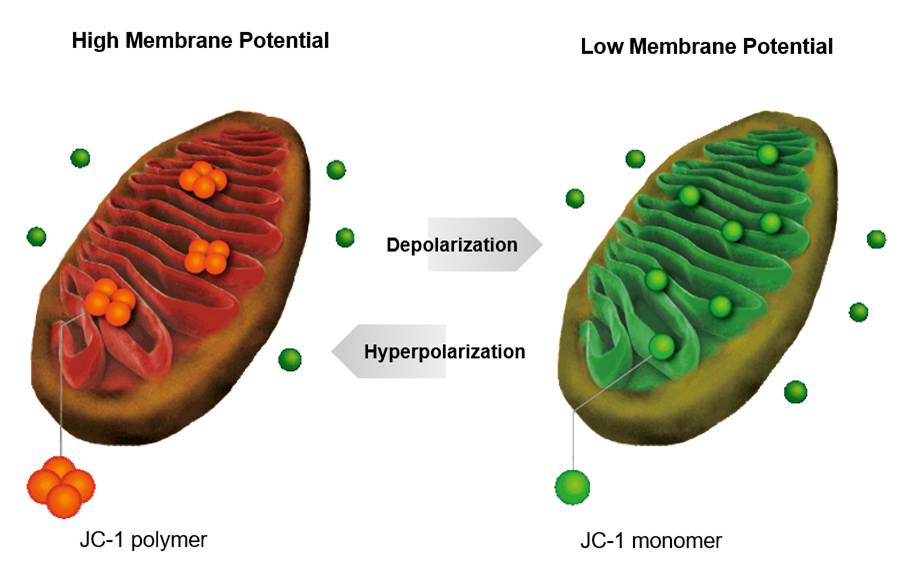
Moreover, CBD and THC decreased invasion by disrupting the mitochondria in the cell. The mitochondria are responsible for respiration to produce energy in the form of adenosine triphosphate (ATP). This in turn increases the accumulation of reactive oxygen species, oxidative stress, and depolarises the mitochondrial membrane potential by the proportion of JC-1 monomers. Please see Figure 3. This was markedly shown at the equimolar concentration of CBD: THC at 2.5:2.5 µM compared to individual treatment. There was a high sensitivity for A2780 (10-fold difference) and SKOV3 (3-fold difference) in mean fluorescence intensity compared to the control. As a result, the number of members of the cell death (apoptosis) that promote apoptosis (pro-apoptotic) increases. For instance, cytochrome C is released from the mitochondria and into the cytoplasm. The molecular scissors (caspases) perform a cascade and initiate apoptosis, which decreases proliferation. This further indicates how CBD and THC can disrupt the mitochondrial membrane potential, which is a biomarker for apoptosis in ovarian cancer cell lines and decrease cellular proliferation (Tong et al., 2025; Boggs et al., 2016).
Further efforts to understand the link between CBD, THC, and their combination with the PI3K/AKT/mTOR signalling pathway and its negative regulator PTEN in ovarian cancer cell lines and their impact on tumour progression and resistance to chemotherapy were achieved (Tong et al., 2025; Boggs et al., 2016).
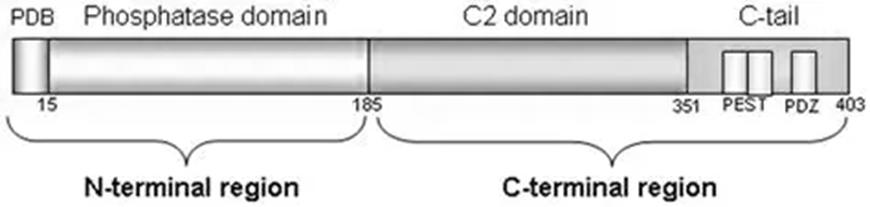
The tumour suppressor PTEN was upregulated when ovarian cells were treated with CBD, THC, or their combination. This was detected particularly in the SKOV3 cell line in the phosphorylation sites of PTEN. Please see the structure of PTEN illustrated in Figure 4. Phosphorylation is the addition of phosphate, and the sites reside in the C-terminus at Ser380, Thr382, and Thr383 in a closed conformation. This upscales the level of stability and limits catalytic activity.
PTEN is a protein composed of 403 amino acids. There are primarily two regions: N-terminal and C-terminal. The N-terminal region is composed of 185 amino acids (positions 1–185) with a PIP2-binding domain (PBD) and a phosphatase domain of 218 amino acids (positions 186–403). The C-terminal region consists of a C2 domain and a tail. The tail comprises two PEST (proline, glutamic acid, serine, threonine) sequences and an ending with a PDZ-interaction motif (Molinari and Frattini, 2014).
On the other hand, dephosphorylation (removal of the phosphate) increased catalytic activity and membrane binding. This highlights how the upregulation of PTEN by CBD, THC, and both antagonised the PI3K/AKT signalling pathway (Tong et al., 2025; Boggs et al., 2016). The signalling pathway is illustrated in Figure 5.
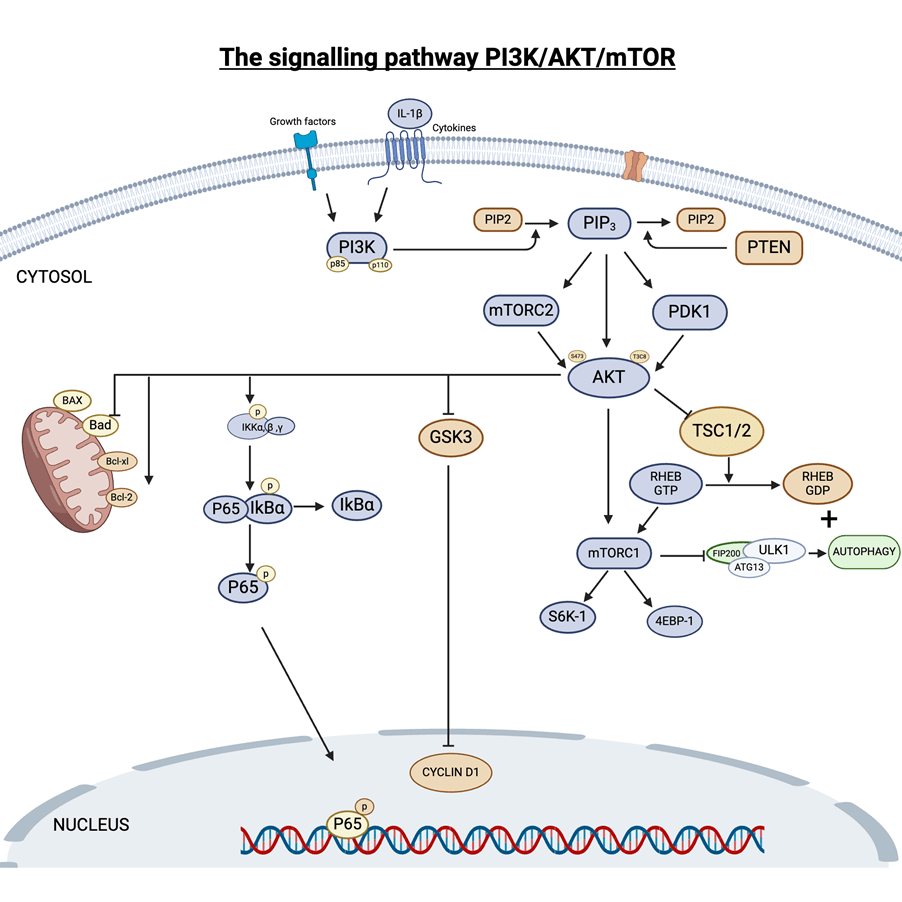
Other proteins, upstream kinases, and phosphatases may have an additional effect of tumour suppression. Casein Kinase 2 (CK2) phosphorylates the C-terminal of the PTEN, whereas PP2A phosphatase dephosphorylates the residues. Oxidative stress and the CB1/CB2 receptor activation by cannabinoids may form post-translational modifications. This consequently downregulates the phosphorylation of p-PI3K, p-Akt, and p-mTOR (Tong et al., 2025; Boggs et al., 2016).
Ultimately, the anti-tumour effects of CBD and THC have been greatly presented mechanistically through in vitro studies. There is a more potent effect as a combined therapy than alone, where it can suppress the PI3K/AKT/mTOR signalling pathway. However, the complexity of the tumour behaviour has not been fully profiled on living organisms. Further studies are needed in vivo to determine its safety before being applied as a patient treatment. Moreover, there is variation in countries for approval as cannabinoid therapy, which may influence translational research and its use as a targeted anti-cancer therapeutic strategy (Tong et al., 2025; Boggs et al., 2016). This is illustrated in Appendix A and B. CBD is available with a doctor’s prescription in most countries.
References
Boggs, D.L., Peckham, A., Boggs, A.A. and Ranganathan, M. (2016). Delta-9-tetrahydrocannabinol and cannabidiol: Separating the chemicals from the ‘weed,’ a pharmacodynamic discussion. Mental Health Clinician, 6(6), pp.277–284. doi:https://doi.org/10.9740/mhc.2016.11.277.
Tong, S., Loilome, W., Namwat, N., Klanrit, P., Wangwiwatsin, A., Win, Z.Z., Koyabuth, P. and Chumworathayi, B. (2025). Selective anti-cancer effects of cannabidiol and Δ9-tetrahydrocannabinol via PI3K/AKT/mTOR inhibition and PTEN restoration in ovarian cancer cells. Frontiers in Pharmacology, 16. doi:https://doi.org/10.3389/fphar.2025.1693129.
Molinari, F. and Frattini, M. (2014). Functions and Regulation of the PTEN Gene in Colorectal Cancer. Frontiers in Oncology, 3. doi:https://doi.org/10.3389/fonc.2013.00326.
Supplements In Review (2017) Is CBD Legal in my Country? Global Guide to CBD Legality. Available at: https://supplementsinreview.com/blog/is-cbd-legal-in-my-country-global-guide-to-cbd-legality/ (Accessed: 20th March 2026).
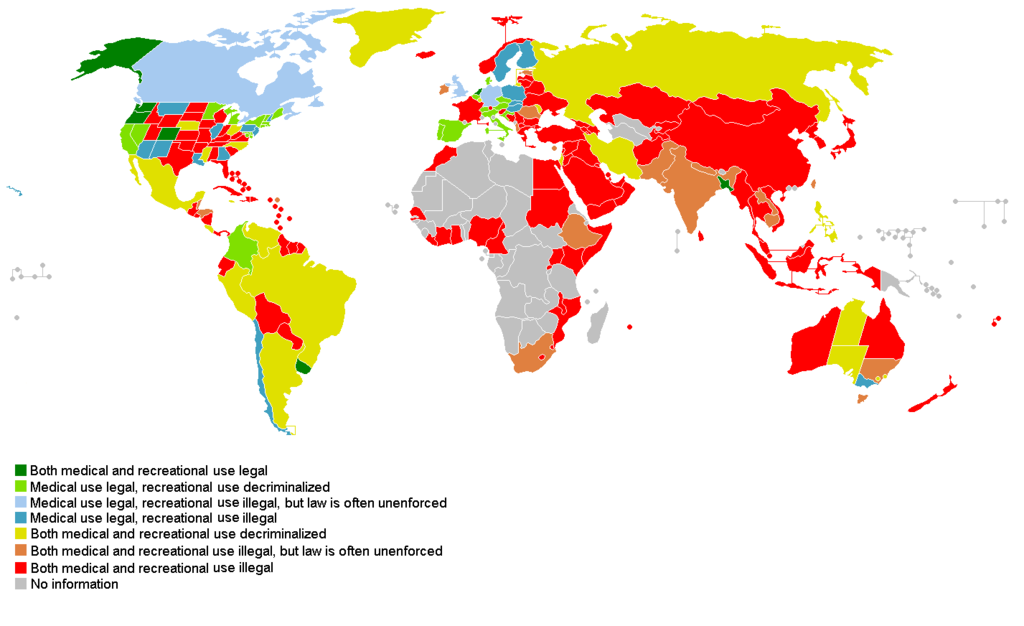
Appendix A: The CBD application status on a global scale (Supplements in Review, 2017)
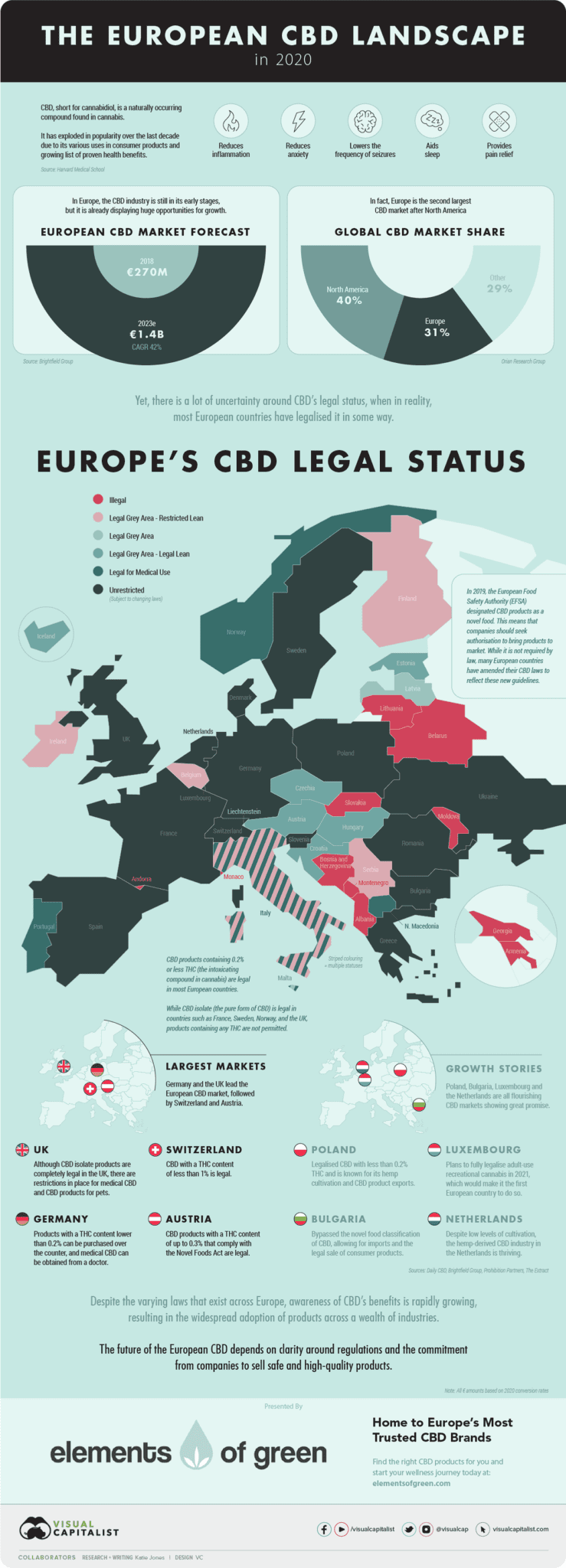
Appendix B: The European application status for CBD use
Leave a comment