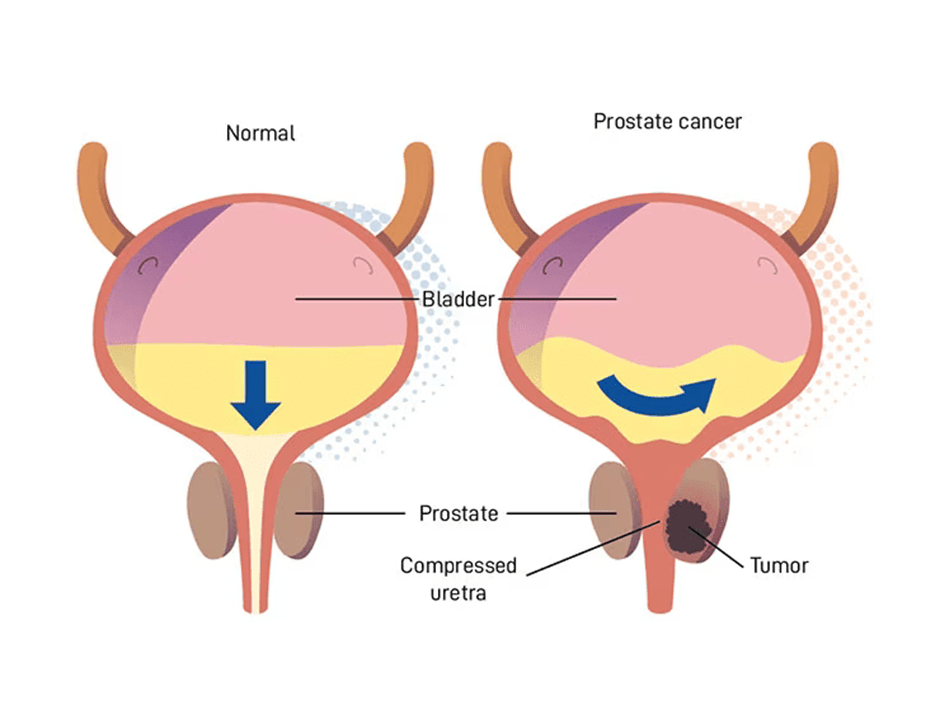
The continual rise of prostate cancer cases worldwide has shifted its ranking from being the second most common cancer to the leading malignancy in males. This corresponds to high morbidity and mortality in recent years (Zannini et al., 2025). The prostate gland is part of the male reproductive system, situated under the bladder, and primarily produces semen, a fluid that contains sperm. Please see Figure 1. At clinical presentation, prostate cancer is heterogeneous, with risk factors ranging from genomic to environmental factors (Evans et al., 2025). This trend is notable in developed countries with longer life expectancy (Zannini et al., 2025). The risk increases with age. About 85% of prostate cancer cases are projected in patients over 60 years old (Zannini et al., 2025). If diagnosed early and localized, prostate cancer has an estimated 99% ten-year survival rate. Active surveillance and curative treatment methods include androgen deprivation therapy (ADT) with radiotherapy for localized disease. Minimally invasive ablative radiotherapy also offers low toxicity (Panebianco et al., 2016). Removal of the prostate (radical prostatectomy), external beam radiotherapy, or brachytherapy are other curative options for localized cancers (Zannini et al., 2025; Panebianco et al., 2016). It is pivotal to find strategies that detect and predict prostate cancer early, reducing progression and improving treatment options (The Institute of Cancer Research, 2025; Zannini et al., 2025).
One of the focused preventative ways established is understanding the genetic variants that cause the subtypes of prostate cancer, whose normal function is involved in DNA damage repair and response (DDR) (Evans et al., 2025). They can also hold prognostic value for targeted therapy, particularly PARP inhibitors. Amongst the genetic risk factors of prostate cancer are BRCA 1/2 and mismatch repair (MMR) genes. Key MMR genes are CHEK2, MLH1, MSH2, MSH6, HOXB13, and PMS2 (Zannini et al., 2025). Randomised clinical trials have indicated that men who have a change in the gene (mutation) of BRCA2 or Ataxia-Telangiectasia Mutated (ATM) have an elevated risk of developing an aggressive (likely to spread) form of prostate cancer and a relatively poor prognosis (Evans et al., 2025).
Alternatively, if a patient has a BRCA1 variant, the lifetime risk of developing prostate cancer rises to some extent (Prostate Cancer UK, 2025). Zannini et al. (2025) revealed that, in some reports, patients with BRCA1 mutations had a 4-fold chance of developing prostate cancer. In comparison, a BRCA2 mutation shows a 3-8.6 fold higher risk. Nevertheless, the evidence remains unclear. Other research claims that having a BRCA1 or BRCA2 gene variant raises the chance of an early prostate cancer diagnosis or of developing more aggressive cancer at advanced stages (Prostate Cancer UK, 2025). Advanced cancers are characterized by metastatic disease, nodal involvement, higher stages T3 and T4, and higher Gleason scores (Zannini et al., 2025).
Recent guidelines have proposed the need for a screening programme after robust clinical evidence. The UK National Screening Committee (UK NSC) has suggested that the target group that needs to be routinely monitored for prostate cancer every two years is men between the ages of 45 and 61 years with a known BRCA1 or BRCA2 genetic variant (Prostate Cancer UK, 2025; Cancer Research UK, 2025; The Institute of Cancer Research, 2025). The aim of this modest review is to analyse the current and prospective diagnostic methods of prostate cancer.
Current Diagnostic Methods
The first diagnostic method following symptom assessment is the digital rectal examination (DRE), where the healthcare professional places a finger up the back passage of the patient to assess whether the prostate gland has increased in size. An enlarged prostate gland (benign prostatic hyperplasia) influences how urine is stored in the bladder and how urine leaves the body via the urethra (micturition). This can be explained by its anatomical position under the bladder.
Further assessment involves the serum measurement of prostate-specific antigen (PSA). The PSA is a protein in the lining of the prostate gland referred to as the glandular epithelium. Elevated levels of PSA beyond 4 ng/ml may correspond to benign prostatic hyperplasia, inflammation (prostatitis), recent ejaculation of semen, DRE, bicycle riding, and older age (Cancer Research UK, 2025). Thus, PSA measurement helps to explore the underlying condition that affects men, transwomen, and non-binary people assigned male at birth (Cancer Research UK, 2025).
Prostate biopsy is subsequently performed for abnormal DRE and high PSA measurement, where a sample of the prostate gland is removed to assess under the microscope whether the tissue is cancerous and, if so, at what stage and grade using the GLEASON score and Cambridge Prognostic group criteria. The cancer stage refers to whether the cancer has spread, whereas the grade distinguishes how different the cells look compared to non-cancerous cells.
However, a profound challenge can be observed where many patients with elevated PSA levels undergo further prostate cancer tests and biopsies that may cause harm because of the overdiagnosis of the slow-growing tumors. It may also miss some prostate cancers (false negatives) and lead to overtreatment with surgery and radiotherapy. These multifactorial reasons can cause side effects, primarily impotence and incontinence. Impotence is where there is difficulty getting an erection, and incontinence, lack of control of the bladder (Institute of Cancer Research, 2025).
In some European countries, it has been recommended that patients with BRCA 1 or 2 mutations, particularly young carriers of the gene, should initiate prostate cancer screening via magnetic resonance imaging (MRI). On the other hand, patients aged 55 years are advised to do PSA testing and multiparametric MRI for accuracy in detection, staging, and active surveillance (Zanneni et al., 2025; Panebianco et al., 2016).
Multiparametric MRI is undertaken in active surveillance for patients who reject biopsy and during biochemical failure. The imaging modality connects T1 and T2-weighted images with a minimum of two functional techniques: dynamic contrast-enhanced imaging (DCEI), MR spectroscopy (MRS), and diffusion-weighted imaging (DWI) (Panebianco et al., 2016). It has high sensitivity and specificity to detect various outcomes post-treatment due to restricted diffusion imaging. where fibrosis and atrophic remnant seminal vesicles (SV) that often appear post-surgery are discovered and are used to differentiate from local relapse (Panebianco et al., 2016). Fibrosis is the scarring of tissue after experiencing injury and inflammation. Patients who had radiotherapy intervention, whether internally or from an external source, can be identified by the decreased signal intensity in the peripheral zone of the T2-weighted image. Common causes are the loss of water content that hypothetically indicates tumours and hemorrhage (Panebianco et al. 2016).
T1-weighted (T1W) and T2-weighted (T2W) MRI have a dual importance. T1 MRI comprises the longitudinal relaxation time (T1). This refers to the time taken for positively charged protons to realign with the external magnetic field after excitation (MRI Master, 2023). It helps to provide anatomical detail of tissues and structures. The fluid-filled structures appear dark and hypointense, whereas fat-rich structures are hyperintense, indicated by the bright colour. A full urinary bladder appears dark, whereas the prostate gland has an intermediate signal in a TW1 imaging (MRI Master, 2023).
On the contrary, T2W presents a pathological update where inflammation, oedema (build-up of fluid), and other lesions (MRI Master, 2023). It corresponds to the variation in the transverse relaxation times (T2) of tissues where excited protons lose phase coherence in response to the interactions with their surroundings. Therefore, the fluid-filled organs are hyperintense (bright); for instance, a full bladder will be bright; however, the T2W signal for the prostate gland is intermediate to bright (MRI Master, 2023).
What Is The BRCA gene?
The BRCA genes are inherited or passed down via our parents, whose normal function is to uphold the growth of normal non-cancerous cells. Under physiological conditions, they correct DNA repair and maintain genomic stability. Amongst the cellular functions are regulating cell cycle, transcription, ubiquitination, homologous recombination, and apoptosis (cell death). Transcription is the initial step in producing proteins, where one of the strands of the DNA is used to make a copy of another type of genetic material referred to as ribonucleic acid (RNA). Ubiquitination is the process by which a small protein called ubiquitin binds and directs non-functional proteins to be broken down in a structure called the proteosome. Homologous recombination shortens DNA regions referred to as nucleotides, which are exchanged between identical DNA molecules or with high similarity to facilitate DNA repair (Zannini et al., 2025).
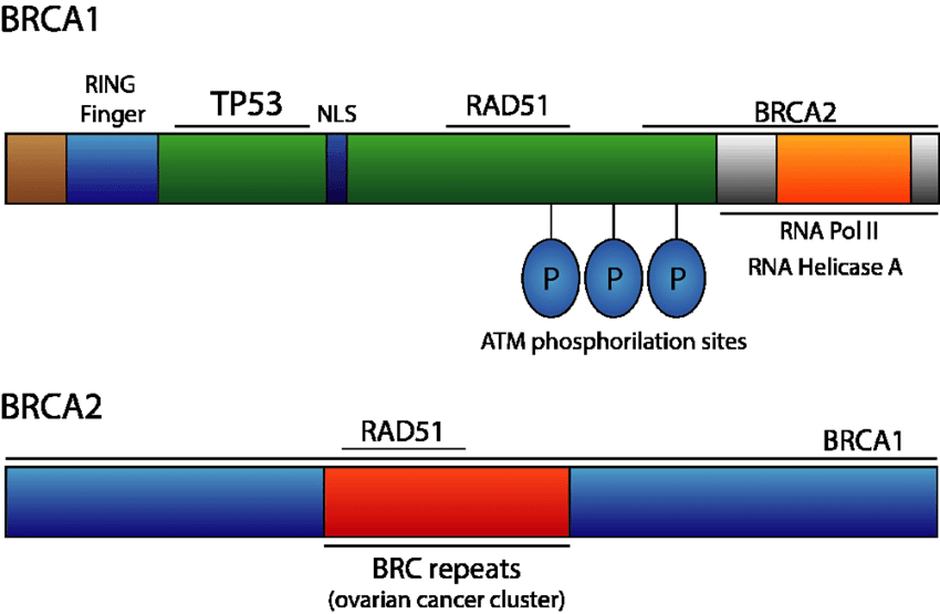
There are two main types: BRCA 1 and 2 (Prostate Cancer UK, 2025). The BRCA1 gene is situated on chromosome 17, whereas the BRCA2 gene is situated on chromosome 13. They vary in the genetic composition, protein structure, and size, as illustrated in Figure 3. The BRCA1 gene consists of 24 exons (coding regions) and encodes a 1836-amino acid nuclear-cytoplasmic transfer protein with a molecular weight of 220 kd. The BRCA1 protein is composed of several domains: ring finger, nuclear localization sequence, coiled-coil (CC), and BRCA1 C-terminus (BRCT). The CC and BRCT interact with other molecules and proteins (Zannini et al., 2025).
The BRCA2 gene consists of 27 exons and encodes a 3418 amino acid nuclear protein that is larger in size; the molecular weight is between 350 and 380 kd. Several domains differ from those of BRCA1. The protein structure consists of a nuclear localization sequence, two functional domains: transactivation and DNA binding domain, and eight BRC-repeat motifs present in the central region whose primary role is in DNA repair (Zannini et al., 2025).
A mutation in the BRCA gene evades the normal function, and the production of these gene variants can be achieved somatically (the body produces abnormal BRCA genes) or inherited from our parents (germline) (Prostate Cancer UK, 2025). It is estimated that one in 300 to 400 people has a BRCA gene variant. Patients from the Ashkenazi Jewish ethnic origin are at higher risk, where around 1 in 40 people have the BRCA mutation (Prostate Cancer UK, 2025). It is important to state that having a BRCA variant and/or relatives with prostate cancer increases the likelihood of one having prostate cancer, but does not suggest that one will have it.
Patients with BRCA1/2 mutations being diagnosed with prostate cancer positively correlate with age, especially after 75 years. The percentage risk for patients with a BRCA1 mutation is 20%; however, after the age of 85 years, it rises to 30%. On the contrary, patients with the BRCA2 mutation after 75 years have 20% higher chance, but this upregulates to 60% after 85 years of age. This indicates how the chance of being diagnosed with prostate cancer increases with the presence of BRCA mutation, but more so with germline BRCA2 pathogenic variants, with over 10% involvement in metastatic disease (Zannini et al., 2025; Evans et al., 2025).
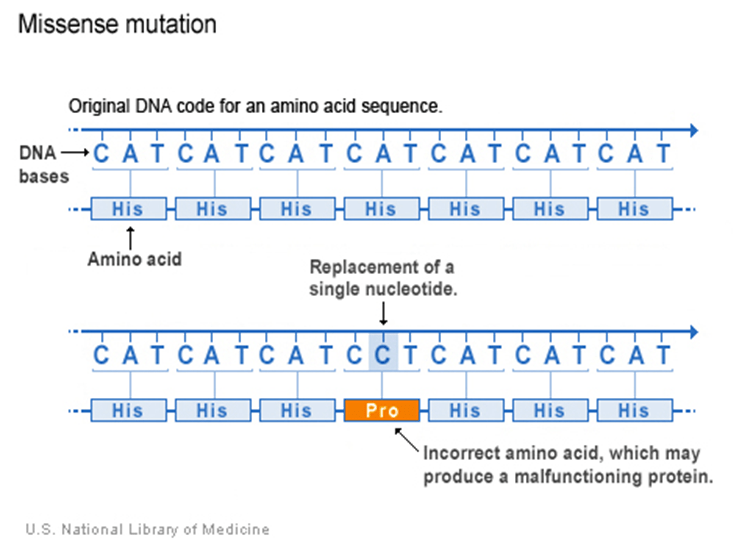
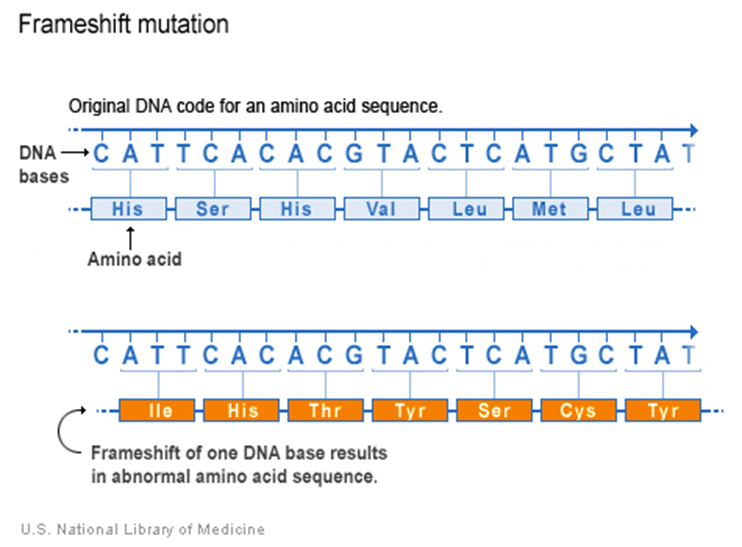
The most frequent mutation in descending order is missense substitution, synonymous substitution, and frameshift deletions. Missense substitution, as portrayed in Figure 4, is when there is a change in a single nucleotide, which alters the amino acid sequence and causes the protein to malfunction. Synonymous substitution is when the variation of the genetic sequence does not alter the amino acid sequence or protein. A frameshift mutation, as pictured in Figure 5, implies that there is an addition or deletion of one nucleotide. Through the integration of Next Generation Sequencing (NGS) technologies, there are five clinically distinctive categories: benign or nonpathogenic, likely benign or likely not pathogenic, uncertain significance, likely pathogenic, and pathogenic. The status of uncertain significance evolves over time as it meets one of the genetic criteria (Zannini et al., 2025).
Moreover, the BRCA genetic variant has a predictive and prognostic value in other hereditary cancers besides prostate: ovarian, breast, and pancreatic (Zannini et al., 2025). Thus, it is essential to assess the level of risk. The National Health Service (NHS) offers a free BRCA gene test for any patient residing in England aged 18 years and above who has one or more Jewish grandparents. A saliva (spit) or blood test can help detect whether one has the BRCA variant (Prostate Cancer UK. 2025).
Other genetically-linked mutations associated with prostate cancer
There are several potential homologous repair genes that correlate with the incidence of prostate cancer: ATM, PALB2, s-CDK12, and Lynch syndrome-associated genes. The NHS germline and somatic testing in patients at risk in secondary and tertiary cancer care are evaluated for the BRCA1, ATM, and CDK12, particularly in patients with metastatic and castrate-resistant prostate cancers (Evans et al., 2025).
Partner and localizer of BRCA2 (PALB2) is a tumour suppressor gene whose primary role in the homologous recombination repair (HRR) pathway is to repair the double-stranded DNA breaks. The incidence risk of prostate cancer with the PALB2 mutation is 12%. There is uncertainty on the role of prostate cancer screening with PSA and MRI in patients who are carriers of the PALB2 pathogenic/likely pathogenic variants (McVeigh, 2022).
Conversely, a pathogenic variant in the ATM gene increases the risk of cancer and a rare genetic condition called ataxia telangiectasia. It is a progressive neurological condition where, during the early onset, there is cerebellothalamic ataxia, conjunctival telangiectasia, chorea, and radiosensitivity (McVeigh, 2025).
The ATM gene encodes for the ATM protein that is involved in maintaining genomic stability in response to double-stranded DNA damage, ionising radiation via the phosphatidylinositol-3 kinase (PIK3)-like serine/threonine kinases. ATR is another phosphatidylinositol-3 kinase (PIK3)-like serine/threonine kinase. The stimulation of ATM and ATR reduces homologous recombination.
It has been reported by McVeigh (2025) that one in 100 people (1%) is likely to have pathogenic and likely pathogenic variants in ATM. The appearance of biallelic variants indicates that patients have pathogenic variants of the ATM gene. Ongoing progress is made to determine how males with ATM pathogenic or likely pathogenic mutations who underwent PSA screening and MRI.
The CDK12 gene encodes the cell cycle-dependent protein kinase, CDK12, that combines with the cyclin K protein. Genetic expression can be established upon the CDK12-Cyclin K complex’s ability to phosphorylate the carboxyl-terminal domain of the enzyme RNA polymerase II (Lu et al., 2024). Other biological functions of CDK12 are the two-step process of protein synthesis: transcription and translation, and pre-mRNA splicing. Its content varies with the type of tissue: the testicles have the highest level of CDK12, whereas the brain has the least.
Evans et al. (2025) analysed 450 tumour DNA samples extracted from formalin-fixed, paraffin-embedded blocks (FFPE). The FFPE samples were obtained from the tumour biopsies that consisted of a minimal tumour content of 20%. Bioinformatic analyses were used to identify sBRCA1/2, sATM, and sCDK12 at variant allele frequencies in localised or metastatic patients in North Western England between the years of 2022 and 2025. There were three types of BRCA1/2 gene testing: germline (n=166), somatic (n=280), and both (n=4) (Evans et al., 2025). The germline testing for BRCA1/2 in the UK was initiated from September 1996 till March 2024. Alternatively, the somatic version for BRCA1/2, ATM, and CDK12 testing took place between January 2021 and March 2024. The application of NGS technologies helped to distinguish the type of mutation. The germline BRCA1/2 and extended panel testing originated from the DNA obtained from peripheral circulating lymphocytes (Evans et al., 2025).
The report findings revealed that from the 450 patients, the BRCA1/2 germline/somatic test had 0.44% (n=2) germline BRCA1 variants compared to 6% germline pathogenic variants in BRCA2 (n=27). There were 6/328 cases that had ATM (1.8%). An overview of Evans et al. (2025) findings is in Figure 6.
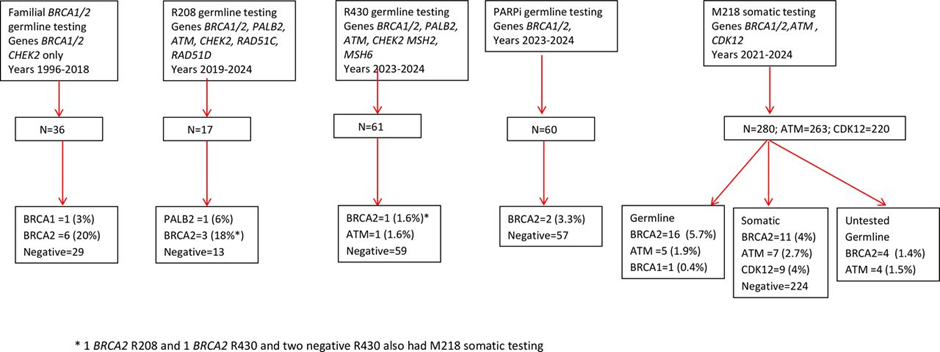
There were 280 patients with metastatic prostate cancer who were examined for somatic, germline, and uncertain mutations. The total pathogenic variants of ATM associated with metastatic disease were 263: Germline (1.9%; n=5), which were confirmed on blood. Somatic (2.7%; n=7), which were confirmed on blood (n=2) and inferred (n=5). There were also samples that had uncertain outcomes (1.5%, n=4). One germline sample was biallelic with variant allele frequencies (VAF) of 58% and 18% (Evans et al., 2025). Individuals who carry pathogenic variants in both copies of their gene are referred to as biallelic variants. Another germline sample had a higher VAF (83%), suggesting loss of wild-type. The four cases of uncertainty had VAF between the range of 37 and 60% (Evans et al., 2025). In addition, 1% of the 97 samples had a single PALB2 pathogenic variant. Therefore, the potential link with prostate cancer is unclear.
However, the total pathogenic variants of CDK12 were 220 and were solely detected in somatic cells (4.1%; n=9). Two samples with CDK12 had biallelic pathogenic variants with low VAF and also had BRCA2 variants. One sample originated from a germline pathogen variant with a VAF of 89%. The other sample had a somatic variant of BRCA2 with a VAF of 13% (Evans et al., 2025). This indicates how CDK12 can exhibit oncogenic features and genetic aberrations in different tumours.
Furthermore, the total pathogenic variants (somatic, germline, uncertain) in BRCA2 were 11% (n= 31/280). There was 4% (n=11) with somatic mutation, 5.7% (n=16) with germline mutations, and uncertain was 1.4% (n=4) in metastatic prostate cancer. Please see Figure 6 of the flow diagram. This is higher compared to BRCA1, which had one variant from each type: germline and somatic in metastatic disease (1/338) (Evans et al., 2025). This illustrates that BRCA2 has a higher mutational frequency than BRCA1.
Some of the findings of Evans et al. (2025) oppose previous studies where Cussenot et al. (2023) revealed BRCA2 and CDK12 somatic pathogenic variants are mutually exclusive. There were two from 220 samples that contained both genes. One sample had a BRCA2 germline variant with biallelic loss of function with a VAF of 89%. The other sample had somatic variants of both genes. In either case, the patients are sensitive to immunotherapy and targeted therapy, for instance, Poly(ADP-ribose) polymerase-1/2 inhibitors (PARPi).
The Test
The UK National Screening Committee (UK NSC) is evaluating a draft recommendation for a potential targeted screening programme that incorporates a public consultation before it is sent to governmental ministers to decide on the programme. The NHS and Health and Social Care system across the UK will then implement the program.
The aim is to invite male patients aged 45 to 61 with a BRCA1 or BRCA2 mutation to be screened every two years for prostate cancer (The Institute of Cancer Research , 2025). Men from the target group will also be offered the PSA test once every two years. Patients with elevated PSA levels will progress to having an MRI scan, whose result will determine whether there is a need to have a biopsy. Patients without high levels of PSA beyond the threshold will be invited in the next PSA test in two years (The Institute of Cancer Research , 2025; Cancer Research UK, 2025).
The prostate screening programme aims to evaluate its cost-effectiveness in preventing false positives leading to mistaken diagnoses and overdiagnosis. Overdiagnosis is where the healthcare professional focuses on slow-growing cancers that cause local forms and or are clinically insignificant prostate cancers (Cancer Research UK, 2025). Thus, there is a dire need to prevent the need to screen all male patients with a family history and people of Afro-Caribbean descent. On the other hand, a potential research question could explore whether Black men of different ethnic backgrounds affect the clinical outcomes. Overall, this helps to identify patients with BRCA-related prostate cancers at an early stage.
Moreover, the Institute of Cancer Research (2025) suggested that from the age of 40, all men with a constitutional genetic variant of BRCA1 or BRCA2 have regular PSA testing, as non-carriers do.
Other supportive evidence are the latest findings from the international IMPACT study revealed that men with BRCA1 or BRCA2 mutations should have an annual measurement test of PSA after it was discovered that males with the BRCA1 gene variant are threefold more likely to have advanced prostate cancer than non-carriers, and this risk is independent of the age factor (The Institute of Cancer Research, 2025). Continuous efforts are made to determine the long-term impact of screening on prostate cancer.
Additionally, patients who are BRCA2 carriers have an increased risk by two-fold from 1.4 to 3.1 with an average age of 60 compared to non-carriers of the BRCA2 variant, whose average age at diagnosis is 65 (The Institute of Cancer Research, 2025). Ongoing trials aim to use a saliva test to detect the BRCA1 gene and PSA, improving early detection.
The BRCA1/2 germline and somatic variants should be tested initially on FFPE and peripheral blood according to the European Society for Medical Oncology (Zannini et al., 2025). They serve as predictive biomarkers for PARP inhibitor therapy, especially olaparib. Peripheral blood allows identification of a constitutional variant. Germline BRCA1/2 via FFPE is used to determine if it is somatic or hereditary in the peripheral blood. This allows family members who are at high risk to undertake genetic counseling to lower risks and preventions.
Limitations In BRCA Screening Performance
There are several technical issues that have arisen with Zannini et al. (2025) that may affect the BRCA testing procedure for somatic testing and not peripheral blood, as follows:
The limited availability of good-quality tumour tissue samples/biomaterial during resection or biopsy
The tumour size.
The percentage of tumour cells present in the sample.
The effect of formalin fixation on DNA fragmentation and yield.
The effect of long-term storage that can degrade the DNA, lowering the DNA concentration and genotyping performance.
Moreover, though analysis of FFPE and peripheral blood is recommended, the rate of failure to detect somatic BRCA1/2 in FFPE varies from 6.7 to 38% in comparison to peripheral blood, with 3%. There are minimal specialised laboratory services that are trained to select tumour areas to enrich neoplastic cells for somatic BRCA testing, and currently, genotyping panels are used to identify mutations.
The aforementioned limitations with FFPE illustrate the significant gold standard procedure for pharmacogenetic analysis of BRCA1/2 mutations in the germline. However, somatic testing cannot be replaced as it facilitates understanding the response to PARP inhibitors.
Ultimately, there are several potential genes that can be elucidated as predictive, prognostic, and diagnostic markers for prostate cancer. However, the presence of BRCA1 and 2 is at a higher degree and has impeded the UK National Screening Committee, which is reviewing the recommendations for the prostate cancer screening programme. Patients with BRCA1 and BRCA2 should be monitored sequentially for early detection and response to PARP inhibitors and other treatments via annual PSA testing. The current method to extract the DNA consists of tumour sample biopsy to form FFPE and peripheral blood. The source of peripheral blood facilitates germline testing (constitutional) in comparison to FFPE. Ongoing efforts are being made to monitor the effectiveness of disease and treatment to prevent evasion of immune surveillance.
References
Cancer Research UK (2025). First steps towards a targeted prostate cancer screening programme. Available at: https://news.cancerresearchuk.org/2025/11/28/uk-nsc-draft-recommendation-targeted-prostate-cancer-screening-programme/ (Accessed: 24th March 2026)
Evans, D.G., Burghel, G., Schlecht, H., Sachdeva, A., Hudson, A., Parikh, O., Lalloo, F., Bristow, R. and Woodward, E.R. (2025). UK-based clinical testing programme for somatic and germline BRCA1/2, ATM and CDK12 mutations in prostate cancer: first results. BMJ Oncology, 4(1), p.e000592. doi:https://doi.org/10.1136/bmjonc-2024-000592.
Godet, I. and M. Gilkes, D. (2017). BRCA1 and BRCA2 mutations and treatment strategies for breast cancer. Integrative Cancer Science and Therapeutics, [online] 4(1). doi:https://doi.org/10.15761/icst.1000228.
Lu, K.-Q., Li, Z.-L., Zhang, Q., Yin, Q., Zhang, Y.-L., Ni, W.-J., Jiang, L.-Z., He, W. and Wang, B. (2024). CDK12 is a potential biomarker for diagnosis, prognosis and immunomodulation in pan-cancer. Scientific Reports, 14(1). doi:https://doi.org/10.1038/s41598-024-56831-7.
McVeigh, T. (2022) PALB2 Available at: https://www.genomicseducation.hee.nhs.uk/genotes/knowledge-hub/palb2/ (Accessed: 24th March 2026)
McVeigh, T. (2025) ATM-associated cancer risk. Available at: https://www.genomicseducation.hee.nhs.uk/genotes/knowledge-hub/atm-associated-cancer-risk/ (Accessed: 24th March 2026)
MRI Master (2023) T1 vs T2 MRI Available at: https://mrimaster.com/t1-vs-t2-mri/#google_vignette Accessed: 24th March 2026
Olivier Cussenot, Géraldine Cancel‐Tassin, S. Madhusudana Rao, Woodcock, D.J., Lamb, A., Mills, I.G. and Hamdy, F.C. (2023). Aligning germline and somatic mutations in prostate cancer. Are genetics changing practice? BJU International. doi:https://doi.org/10.1111/bju.16120.
Panebianco, V., Barchetti, F., Grompone, M.D., Colarieti, A., Salvo, V., Cardone, G. and Catalano, C. (2016). Magnetic resonance imaging for localization of prostate cancer in the setting of biochemical recurrence. Urologic Oncology: Seminars and Original Investigations, 34(7), pp.303–310. doi:https://doi.org/10.1016/j.urolonc.2016.01.004.
Prostate Cancer UK (2025) BRCA genes and prostate cancer risk. Available at: https://prostatecanceruk.org/prostate-information-and-support/risk-and-symptoms/are-you-at-risk/brca-genes-and-prostate-cancer-risk (Accessed: 24th March 2026)
The Institute of Cancer Research (2025) Men with BRCA1 and BRCA2 gene mutations should get annual prostate cancer screening. Available at: https://www.icr.ac.uk/about-us/icr-news/detail/men-with-brca1-and-brca2-gene-mutations-should-get-annual-prostate-cancer-screening (Accessed: 24th March 2026)
Zannini, G., Facchini, G., De Sio, M., De Vita, F., Pagliuca, F., Franco, R. and Zito Marino, F. (2025). BRCA1 and BRCA2 mutations testing in prostate cancer: Detection in formalin fixed paraffin embedded (FFPE) and blood samples. Pathology – Research and Practice, [online] 266, p.155803. doi:https://doi.org/10.1016/j.prp.2024.155803.
Zhang, Y., Wells, S., Roldan-Alzate, A. and Hernando, D. (2019). Simultaneous T1, T2 and ADC Mapping in Prostate Cancer and BPH using Stimulated-Echo based Mapping (STEM). Proceedings of the International Society for Magnetic Resonance in Medicine. 27
Leave a comment