Prostate cancer is the most common cancer that affects the male population. National Institute for Health and Care Excellence (2026) revealed that prostate cancer accounted for 46000 cases in England and Wales between 2019 and 2020. 13% of these cases have progressed to metastasis at diagnosis. There are ca. 12,000 deaths linked to prostate cancer annually (National Institute for Health and Care Excellence, 2026).
There are ongoing efforts to improve diagnosis and develop therapeutic strategies to enhance overall survival. Deprivation of the male hormones (androgens) is the prominent treatment for advanced-stage cancers and can be surgically established via medical castration. This decreases the synthesis of the male-dominant steroid hormone, testosterone, and its derivative dihydrotestosterone (DHT). This form of prostate cancer is referred to as castrate-resistant prostate cancer (CRPC). The addition of androgen antagonists to androgen deprivation has facilitated decreasing the concentration of prostate-specific antigens (PSA) for a short-term effect rather than a prolonged option. The PSA is a serum biomarker where elevated levels may signify prostate cancer or prostate-related conditions. Recently, the hormonal drug, Abiraterone, has proven both therapeutic and cost-effective value. The NHS England (2026) has announced that more than 2000 patients with early-stage prostate cancer will have access to the drug. An additional 7000 men with high-risk prostate cancer estimated annually will be eligible for the drug (NHS England, 2026; Gunn, 2026).
Moreover, in recent years, the poly ADP-ribose polymerase (PARP) inhibitors have been the first class of targeted therapies and have shown clinical benefit. They are applied as a standard therapy for mCRPC patients with defective Breast Cancer Susceptibility Gene 1 (BRCA1) and Breast Cancer Susceptibility Gene 2 (BRCA2) and homologous recombination repair (HRR) gene germline and somatic mutations. The emergence of the somatic BRCA1/2 mutations facilitates the use as predictive markers for PARP inhibitors, whereas germ-line BRCA1/2 mutations are identifiable in multiple hereditary cancers, facilitating their use as a prognostic factor to assess risk and guide preventative strategies (Zanini et al., 2025). The targeted PARP inhibitors aim to evade the DNA repair mechanisms and induce apoptosis (cancer cell death) (Zannini et al., 2025; Evans et al., 2025). This article aims to explore the therapeutic effects of Abiraterone, PARP inhibitors, and other targeted approaches to maximise and improve the quality of life for patients with prostate cancer.
The Function Of Androgens
Testosterone and DHT are produced in the Leydig cells of the testicles, which account for about 95% of androgen production in males. However, alternative sources such as the adrenal glands and the prostate gland can also produce androgens, allowing cancer growth to continue even when androgen levels are reduced to castrate levels (Rehman and Rosenberg, 2012). Testosterone is also found, though in smaller quantities, in the ovaries of females (MyEndoConsult, 2026).
The gonads, testicles and ovaries are essential for the production of germ cells, sperm and ova/eggs respectively. This process is referred to as gametogenesis. The two primary roles of the gonads are to secrete steroid sex hormones that facilitate the growth, functionality, and maintenance of the reproductive organs and the development of secondary sexual characteristics (MyEndoConsult, 2026). The testicles are in a constant position of producing sperm and testosterone from ca. 13 years old in puberty until the endpoint (senescence). This portrays the importance of the testicles metaphorically as a maestro to produce sperm (spermatogenesis) and hormones (MyEndoConsult, 2026).
DHT plays a key role in the differentiation of male external genitalia and the prostate gland during fetal development. It also drives the growth of the prostate gland and penis at puberty, leading to characteristics such as facial hair, acne, and scalp hair recession in adult life. In contrast, testosterone is essential for developing the Wolffian ducts into male internal genitalia during fetal development.
The Androgen Receptor Signalling Pathway
Several contemporary studies have demonstrated how the stimulation of the androgen receptor (AR) can increase the growth of prostate cancer. Abiraterone stops the body from producing testosterone by halting the enzyme, cytochrome P450 16 alpha-hydroxylase (CYP17) as presented in Figure 1 (Rehman and Rosenberg, 2026). It highlights being more effective in hormone-sensitive prostate cancers that rely on testosterone.
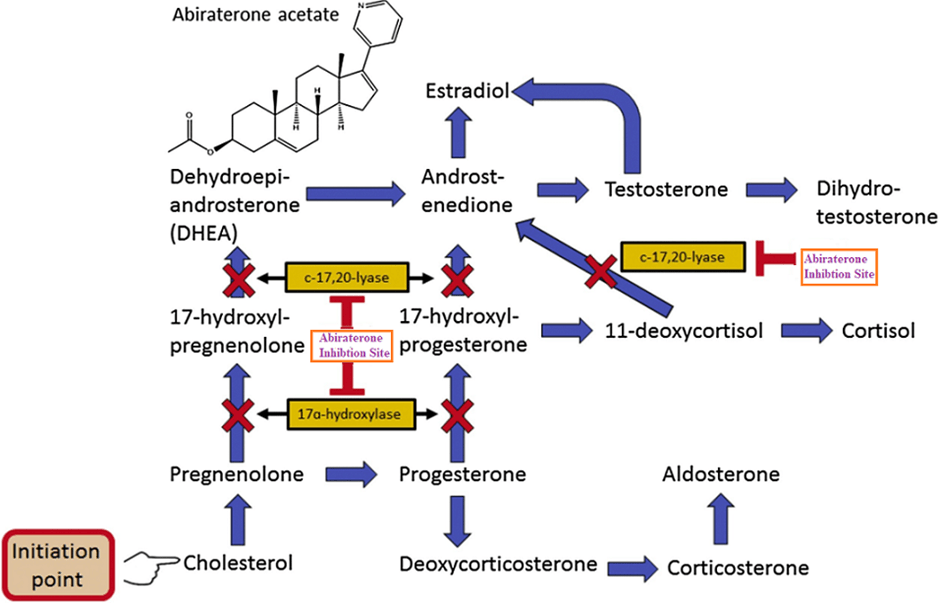
The CYP17 enzyme is situated on the endoplasmic reticulum in several organs: testicles, adrenal glands, ovaries, and the placenta. Its activity is composed of the combination of 17α hydroxylase and C17,20 lyase. Thus, it is integral in the production of glucocorticoids and sex hormones and is regulated by cytochrome b5. Cortisol is an example of a glucocorticoid hormone that is released during stress conditions, exerting multiple physiological effects on the body: fluid retention, increased sodium levels (hypernatraemia), high blood pressure (hypertension), and low levels of potassium in the blood (hypokalaemia), lowers bone formation, suppresses the immune system, and stimulates sugar level (glucose and glycogen turnover). A low ratio of b5/c lyase facilitates androgen production in the adrenal glands, whereas a high b5/CYP 17,20 in the testicles can elevate the production.
It is estimated that 98% of testosterone in the blood plasma is associated with proteins: sex hormone binding globulins (SHBG), and the remainder is bound to albumin and other transport proteins (MyEndoConsult, 2026). The SHBG protein is a byproduct of liver synthesis. Some unbound or biologically active testosterone is transported freely in serum blood but has a minimal lifespan of 10 to 20 minutes (Rehman and Rosenberg, 2026). The free testosterone is converted to dihydrotestosterone by the enzyme 5α-reductase in the testes. Other free testosterone is metabolised into non-reactive 17-ketosteroids and inert polar metabolites. These products are rapidly excreted in the urine (MyEndoConsult, 2026).
On a cellular level, androgen production is mediated by the androgen receptor. The structure of the androgen receptor and its signalling pathway is illustrated in Figure 2. The receptor consists of several domains: amino-terminal activating domain (NTD), DNA-binding domain (DBD), hinge region, and a carboxyl-terminal ligand-binding domain (LBD). The inactive form of the androgen receptor is bound to heat shock proteins (Hsp) in the cytoplasm. The cytoplasm is a space inside the cell where chemical reactions take place. The transcription factor (androgen receptor) becomes activated when it connects with the testosterone derivative dihydrotestosterone (DHT) in the cytoplasm of the cell. DHT has a two-fold higher affinity than testosterone and a slower dissociation rate (MyEndoConsult, 2026). This causes the dissociation between Hsp and AR. The AR-DHT complex undergoes phosphorylation and translocates to the nucleus, where it renders expression of target androgen-dependent genes by binding to specific Androgen Response Elements (ARE) in the DNA. Examples of target genes are PSA (KLK3) and TMPRSS2 that encode for proteins involved in cell proliferation and survival (Miller et al., 2023; Rehman and Rosenberg, 2012). An alternative schematic diagram is presented in Figure 3.
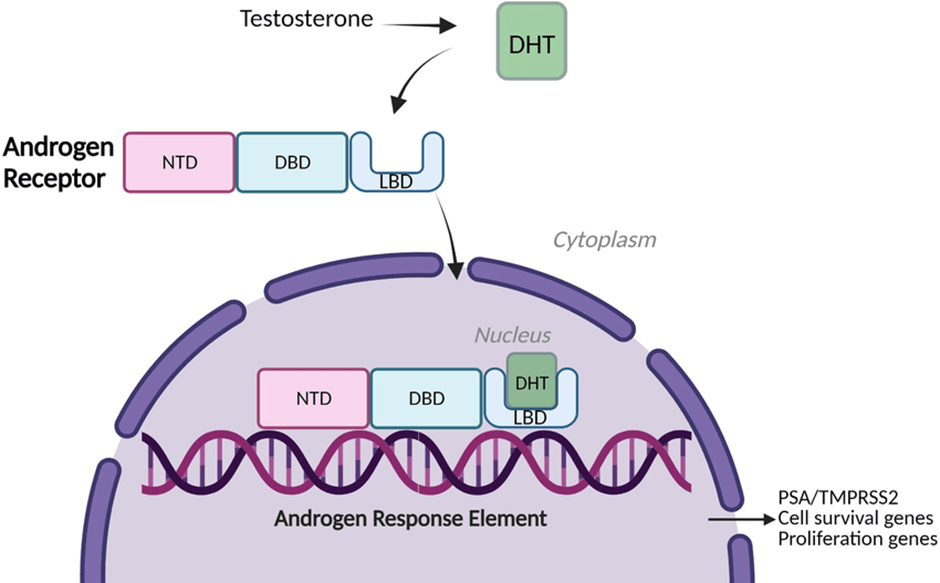
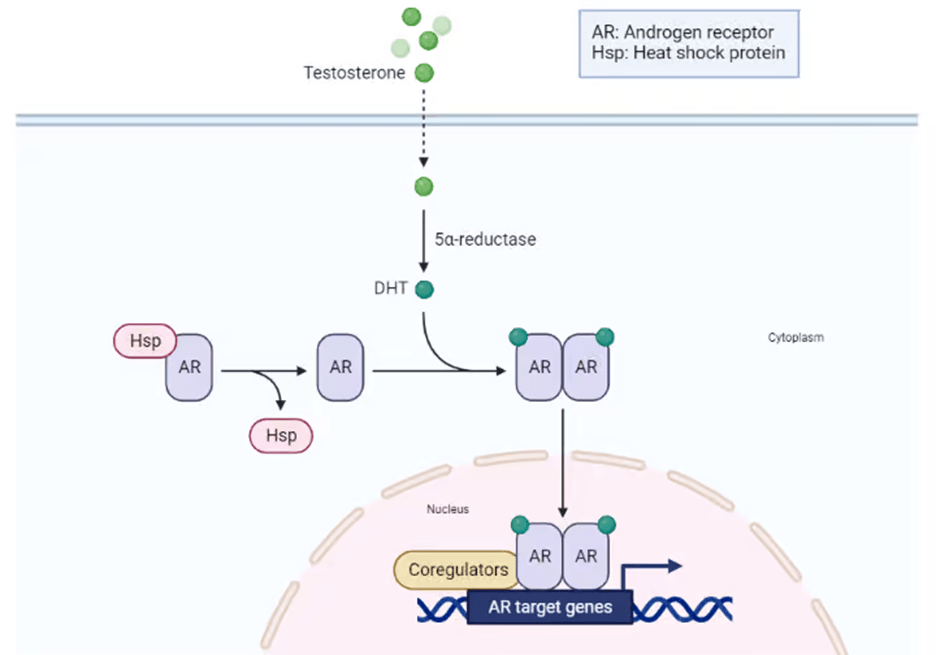
Dysregulation of the androgen signalling pathway can cause castration resistance in patients with prostate cancer. This originates in the mutation of the androgen receptor, gene amplification (copies), upregulation of nuclear transcription coactivators, and the fusion of the target gene TMPRESS2 with the ETS family of oncogenes, ERG or ETV1, though there are low levels of circulating androgens or with relatively low potent androgens (Rehman and Rosenberg, 2012). Mutations in the AR can cause a loss of specificity to androgens. Thus, the AR becomes more responsive towards antiandrogens, for instance, bicalutamide, but not flutamide. This indicates there is a form of selective pressure. Mutational AR may also signal with female hormones (oestrogen and progesterone). This leads to increased prostate cancer growth, though the presence of castrate levels of testosterone is shown (Rehman and Rosenberg, 2012). Another suggestive mechanism is how prostate cancer cells can maintain androgens by self-producing steroid hormones (intratumoral steroidogenesis) to facilitate tumour growth.
Moreover, the halting of androgen synthesis via CYP17 by abiraterone leads to the accumulation of androgens and can possibly activate an altered androgen receptor. This may explain why low levels of prostate-specific antigen (PSA) had a short-term effect upon treatment with abiraterone, where it later markedly increased. A possible explanation may also be linked to the AR having a high affinity for cortisol. Further studies are needed to explain the mechanism (Rehman and Rosenberg, 2012).
About Abiraterone
Abiraterone was developed in the 1990s and was first approved for treatment in the early 2010s for advanced prostate cancers (Gunn, 2026). It stops the production of testosterone and is distinct from other hormone therapies that target the male hormone because it is the sole drug that stops the hormonal synthesis by the cells themselves. This lowers the chances of abiraterone resistance in patients with prostate cancer (Gunn, 2026).
The addition of the steroid, prednisolone, to Abiraterone with androgen deprivation therapy (ADT) helps to lower side effects. However, patients with metastatic prostate cancer who are starting prolonged long-term ADT should not have combined treatment of abiraterone and the second-generation androgen receptor antagonist enzalutamide, nor concomitant use with steroids (Zannini et al., 2025).
The recent results from the phase 3 STAMPEDE trial, which began in 2005, were to evaluate the combined effectiveness of various types of drugs in combination with hormone therapy for the treatment of high-risk prostate cancer localised in the prostate gland and not spread to other parts of the body. Amongst the proposed treatments investigated was abiraterone, where they discovered the risk of recurrent cancer decreased by 50% and the death rate by 40% (Gunn, 2026). Patients have shown improved survival rate after six years with abiraterone (86%) compared to standardized treatment of hormone therapy with or without radiotherapy (77%) (NHS England, 2026). The combinatorial therapy of abiraterone, hormone therapy, and radiotherapy could also be a potential treatment option for some patients. The NHS regions, Wales and Scotland, have offered abiraterone more widely in 2023, and now NHS England has given the green light and approved its use. The decision was made after the cost of buying and delivery of the generic medicine was within budget. The policy to commission abiraterone took place in December 2024, and the National Institute for Health and Care Excellence (NICE) in 2025 recommended it in November 2025 (NHS England, 2026). The NHS England (2026) has set a target to save more than £1 billion pounds on clinically effective biosimilar drugs. Currently, more than eight in ten drugs are prescribed at a lower cost because the manufacturing of biosimilar drugs has been established.
Targeted therapy: PARP inhibitors
The two poly ADP-ribose polymerase (PARP) inhibitors (PARP) approved for the treatment of mCRPC with BRCA1/2 somatic and germline mutations post previous treatment via the Food and Drug Administration (FDA) is Rucaparib and Olaparib (Zannini et al., 2025). Another recommended PARP inhibitor by NICE is talazoparib (National Institute of Health Care and Excellence, 2026). It is given in conjunction with enzalutamide in patients with advanced prostate cancer. The combined treatment is administered to patients who cannot tolerate or are not eligible to take abiraterone and prednisolone, nor chemotherapy. This allows more treatment options, giving flexibility, and there are ca. 2400 people who are eligible for Talazoparib. Clinical trials revealed that the overall survival for Talazoparib with enalutamide is 45.8 months compared to 37 months for patients who had enzalutamide alone. Moreover, it delayed the onset or cancer progression from 33.1 months for combined treatment than enalutamide alone for a duration of 19.5 months (National Institute of Health and Care Excellence, 2026). The time intervals for overall survival and progression-free survival indicate the potent effects of PARP inhibitors with other standard therapies.
The primary role of PARP is DNA repair, particularly double-strand DNA breaks (DSB) and single-stranded DNA breaks (SSB). Please see Figure 4. There are two pathways that repair DSB that target specific phases of the cell cycle. The homologous DNA recombination (HRD) repair is induced by a donor DNA insertion that leads to a gene being inserted or replaced. This causes gene knock-in (Kadam, Lee, and Dhiman, 2023). The HDR repair activates the S/G2 phases of the cell cycle because of the presence of a sister chromatid (Rehman and Rosenberg, 2012).
In contrast, the nonhomologous end joining (NHEJ) repair DSB without any donor DNA. The presence of indel mutations and gene mutations causes gene knock-out (Kadam, Lee, and Dhiman, 2023). This is achieved through the cell cycle, particularly the G1 phase, with the exception of mitosis (Rehman and Rosenberg, 2012). Mitosis is a type of cell division that aids in the growth and repair of cells.
Recent new proteins that facilitate NHEJ are PAXX, IFFO1, TARDBP of TDP-43, MRI/CYREN, RNase H2, and ERCC6L2. Previously known proteins that facilitate NHEJ are DNA polymerase λ/μ, Ku70/80, DNA-PKcs, XLF, and Artemis (Zannini et al., 2025). This demonstrates the plethora of target proteins in DNA repair mechanisms. On the contrary, some target genes act as a double-edged sword where they facilitate NHEJ in G1 but prevent NHEJ in S and G2 phase (Zannini et al., 2025).
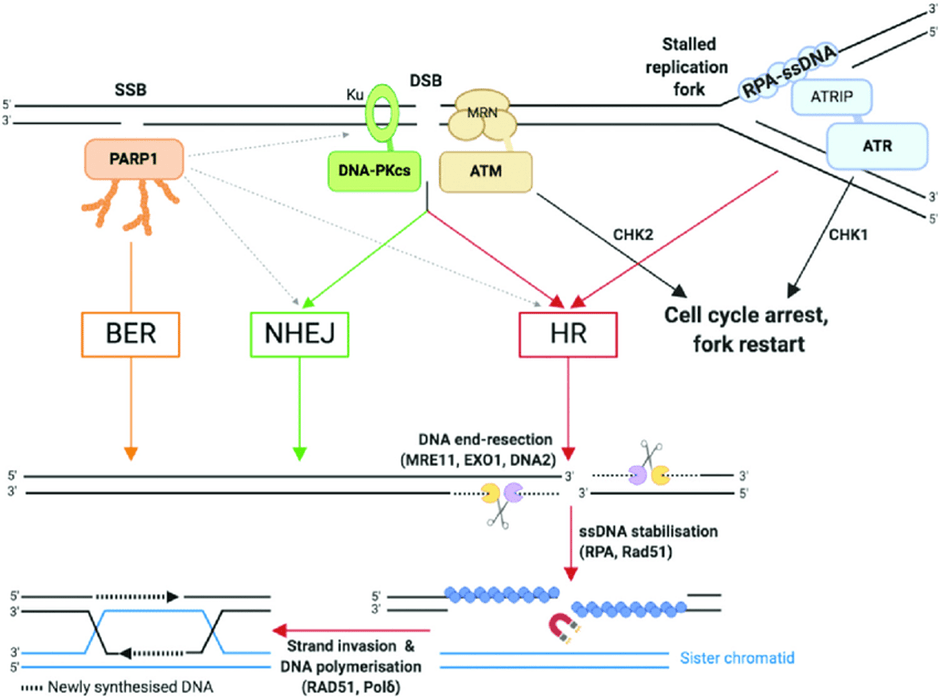
Furthermore, Evans et al. (2025) have revealed there are other mutations that have a role in the pathophysiology of prostate cancer are being explored for their response to targeted inhibitor therapies. For instance, mutated ataxia telangiectasia (ATM) in metastatic prostate cancer are being investigated for rad3-related inhibitor therapy. Patients with CDK12 biallelic loss or deficiency are also characterised with gene fusions and focal tandem duplications which strenghtnes the immune response and can be antagonised with immune checkpoint inhibitors (Evans et al., 2025).
Targeted therapy VIR 5500
Early clinical trials revealed that the immunotherapy VIR 5500 can decrease the size of tumours in patients with advanced prostate cancers. The VIR 5500 can be activated inside the tumour cells and functions as an engineered antibody that accumulates and engages the T killer cells. The phase 1 clinical trial discovered that 58 men with advanced prostate cancers who paused or stopped other treatments were given VIR-5500 (Davis, 2026). Most of the patients (88%) had mild side effects towards the treatment. It was given at low doses and concentration increased gradually. Subjects who administered the highest dose (n=17) had variable response towards their PSA level that markedly decreased. Some subjects (80%; n=14) had their PSA level decreased by 50%. There were nine subjects (53%) whose PSA level fell by 90% and five (29%) had ca. 99% (Davis, 2026). Tumour shrinkage was observed in 5 of the 11 patients who had the highest dose and their tumours csan be measured. However these results are just the start and results are yet to peer-review (Davis, 2026).
Stereotactic Ablative Radiotherapy With Androgen Deprivation Therapy (ADT)
Patients with localised hormone-sensitive prostate cancers have presented high levels of survival with combined therapy. This is facilitated by the robus evidence of the activation of AR to improve DNA repair. To optimise SABR planning, luteinizing hormone-releasing hormone antagonist is given six months from Day 0 of SABR. Personalised medicine is needed because advanced prostate cancers are castration-reisstant where they keep on changing despite having ADT that can be overcome by biomarkers that monitor metastasis and prognosis post-treatment (Zannenni et al., 2025).
Ultimately, there has been mass improvement in developing treatments for patients with mCRPC. Darolutamide, abiratone and Talazoparib function effectively in hormone-sensitive advanced prostate cancers. This helps to decrease testosterone and its application has elevated because of the manufacturing of biosimilars that are deemed cost effective and minimal side effects. Personalised medicine is pivotal for a positive clinical outcome in the treatment of prostate cancer patients.
References
Davis, N. (2026) Researchers praise ‘stunning’ results of new prostate cancer treatment. Available at: https://www.theguardian.com/society/2026/feb/28/researchers-praise-stunning-results-of-new-prostate-cancer-treatment (Accessed: 25th March 2026).
Evans, D.G., Burghel, G., Schlecht, H., Sachdeva, A., Hudson, A., Parikh, O., Lalloo, F., Bristow, R. and Woodward, E.R. (2025). UK-based clinical testing programme for somatic and germline BRCA1/2, ATM and CDK12 mutations in prostate cancer: first results. BMJ Oncology,
Gunn, T. (2026) Thousands more men in England offered abiraterone, one of our prostate cancer treatment breakthroughs. Available at: https://news.cancerresearchuk.org/2026/01/16/thousands-more-men-in-england-offered-abiraterone-prostate-cancer-treatment/ (Accessed: 24th March 2026).
Kadam, S., Lee, D. and Dhiman, P. (2023). Genome-Editing Approaches for Biofortification of Cereal Crops (eds) Biofortification in Cereals. Springer Link. pp.93–126. Available at: doi:https://doi.org/10.1007/978-981-19-4308-9_4.
Miller, K.J., Henry, I., Maylin, Z., Smith, C., Arunachalam, E., Pandha, H. and Asim, M. (2023). A compendium of Androgen Receptor Variant 7 target genes and their role in Castration Resistant Prostate Cancer. Frontiers in Oncology, [online] 13, p.1129140. doi:https://doi.org/10.3389/fonc.2023.1129140.
MyEndoConsult (2026) Male Reproductive Health System. Available at: https://myMyEndoConsult.com/learn/male-reproductive-health-system/ (Accessed: 25th March 2026)
National Insititute of Healthcare and Excellence (2026) Thousands could benefit from new take-at-home treatment for advanced prostate cancer. Available at: https://www.nice.org.uk/news/articles/nice-recommends-new-take-at-home-treatment-for-thousands-with-advanced-prostate-cancer (Accessed: 25th March 2026).
NHS England (2026) NHS to offer thousands of men life-extending prostate cancer drug. Available at: https://www.england.nhs.uk/2026/01/nhs-to-offer-thousands-of-men-life-extending-prostate-cancer-drug/ (Accessed: 24th March 2026)
Rehman, Y. and Rosenberg, J.E. (2012). Abiraterone acetate: oral androgen biosynthesis inhibitor for treatment of castration-resistant prostate cancer. Drug Design, Development and Therapy, 6, p.13-18 doi:https://doi.org/10.2147/dddt.s15850.
Zannini, G., Facchini, G., De Sio, M., De Vita, F., Pagliuca, F., Franco, R. and Zito Marino, F. (2025). BRCA1 and BRCA2 mutations testing in prostate cancer: Detection in formalin fixed paraffin embedded (FFPE) and blood samples. Pathology – Research and Practice, [online] 266, p.155803. doi:https://doi.org/10.1016/j.prp.2024.155803.
Zell, J., Francesco Rota Sperti, Britton, S. and Monchaud, D. (2021). DNA folds threaten genetic stability and can be leveraged for chemotherapy. RSC chemical biology, 2(1), pp.47–76. doi:https://doi.org/10.1039/d0cb00151a.
Leave a comment